Antiphospholipid syndrome testing kit
The BioPlex 2200 Antiphospholipid Syndrome (APLS) IgG, IgM and IgA kits allow for detection of multiple antibodies, aiding in the diagnosis of primary and secondary Antiphospholipid Syndrome (APS). With assay cutoffs set at the 99th percentile in normal subjects, as described by the 2006 Miyakis/Sapporo criteria of APS, the BioPlex 2200 APLS assays offer high specificity, minimizing false-positive rates. The assays offer total %CVs of less than 10.7% for all six. The BioPlex 2200 System can generate up to 200 APS test results per hour, eliminating the need for batched runs. BioPlex 2200 APLS assays include: anti-Cardiolipin IgG, IgM and IgA, as well as anti-Beta-2 Glycoprotein I IgG, IgM and IgA. Bio-Rad Laboratories
ELISA Borrelia test system
The Zeus ELISA Borrelia VlsE1/pepC10 IgG/IgM test system is intended for the qualitative direction of IgG and IgM class antibodies to VlsE1 and pepC10 antigens from Borrelia burgdorferi in human serum. The assay is intended for testing serum samples from symptomatic patients or patients who have a history of Lyme Borreliosis. Independent research has shown (http://www.ncbi.nlm.nih.gov/pubmed/12695997) that the combination of these two antigens produces an assay with clinical sensitivity and specificity, reducing lab costs for confirmatory tests such as the Western Blot. The test system includes SAVe Diluent, which changes color when serum is added, ensuring result integrity and eliminating errors from missed samples. The test is capable of being run in a manual, semi-automated, or fully automated capacity. Zeus Scientific
Anti-CCP test
Siemens Healthcare Diagnostics' FDA-cleared anti-cyclic citrullinated peptide (anti-CCP) IgG assay to aid diagnosis of rheumatoid arthritis (RA) is now offered to U.S. laboratories. Available on Siemens IMMULITE 2000/2000 XPi immunoassay systems, the anti-CCP IgG assay affords laboratories the ability to integrate RA testing onto an automated, random-access analyzer. Currently, the anti-CCP antibody is considered the most specific RA marker and is critical for early diagnosis. With a clinical specificity of 97%, the IMMULITE anti-CCP IgG assay offers laboratories and clinicians a highly accurate diagnostic tool for RA diagnosis. Siemens Healthcare Diagnostics
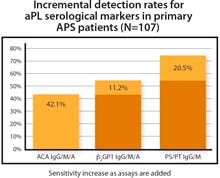
Detection of antiphospholipid syndrome
The QUANTA Lite PS/PT IgG & IgM is designed for the detection of serological markers for Antiphospholipid Syndrome (APS). Antibodies to the complex of phosphatidylserine/prothrombin (PS/PT) provide important information for clinical decision making. Running QUANTA Lite PS/PT along with beta-2-glycoprotein-1 and anticardiolipin tests significantly increases sensitivity for detection of primary APS. Moreover, positivity for PS/PT can aid in risk stratification. Patients with positive results on multiple APS markers are at a far greater risk of thrombosis and pregnancy complications than patients with a single positive serological test for APS. Inova Diagnostics
About the Author
Sign up for our eNewsletters
Get the latest news and updates