Clinical Chemistry analyzer
The BS-200 is a bench-top, discrete and random access clinical Chemistry analyzer with 200 tests per hour; effective for small laboratories, used as a main instrument, or medium/big laboratories, utilized as a back-up instrument. The FDA-cleared model provides laboratories with a broad Chemistry assay menu and real-time sample reaction status. The BS-200 has the ability to analyze up to 330 tests per hour with ISE (K, Na & Cl).The analyzer has a 24-hour refrigeration sample/reagent carousel, built-in bar code scanner, and bi-directional LIS interface. The device employs user-friendly operation software, along with automatic calibration (ISE) and rerun. The BS-200 features pre-dilution and post-dilution for samples, an independent mixing stirrer, and disposable cuvettes. It requires no hands-on maintenance and has a mean time between failures of twelve months.Mindray
Blood lead testing
Magellan Diagnostics introduces LeadCare Ultra, a quantitative, FDA (510k) cleared, bench-top solution for laboratory blood lead testing. The six-channel analyzer allows stat testing with results in just three minutes, and batches at a rate of 90 tests per hour. Its closed-system design requires no set-up or routine maintenance costs, and features electronic calibration, a barcode scanner, QC tracking, and LIS connectivity. The LeadCare Ultra system is based on technology that has been in clinical use for more than two decades. Magellan also offers a portable, CLIA-waived blood lead analyzer, the LeadCare II. Magellan Diagnostics, Inc.
Flexible tissue processor
The LOGOS is the all-in-one tissue processor with the exclusive Milestone patented microwave hybrid processing technology; addressing the needs of "lean" labs for a flexible tissue processor, LOGOS is highly adaptable to customized workflow streams. The LOGOS can operate as a conventional resistance heated overnight tissue processor. Ultra-rapid dehydration/clearing steps are carried out by microwave heating technology for faster turnaround. The retort’s resistance heating speeds up the ramp-up time to the pre-set processing temperature. Features include fully automated processing in overnight and rapid mode, parallel processing in continuous flexible-size batch mode, up to 140 cassettes with 1-gallon standard reagent containers, up to 210 cassettes with 5-liter reagent jars. The LOGOS can process any type of tissue, up to 6 mm thickness. The processors are green, safe and economical; there is no transfer of reagents to tanks. Milestone Medical
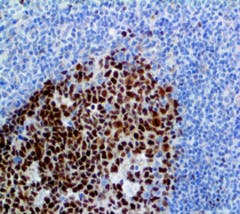
Rabbit monoclonal antibody for IVD use
The Bcl-6 is a transcriptional regulator gene which codes for a 706-amino-acid nuclear zinc finger protein. Antibodies to this protein stain the germinal center cells in lymphoid follicles, follicular cells, and interfollicular cells in follicular lymphoma, large B-cell lymphomas, and Burkitt’s lymphoma, and the majority of the Reed-Sternberg cells in nodular lymphocyte-predominant Hodgkin’s disease. Bcl-6 is also useful in identifying neoplastic cells in cases of nodular lymphocyte-predominant Hodgkin’s disease. In contrast, anti-Bcl-6 rarely stains mantle-cell lymphoma and MALT lymphoma. Bcl-6 expression is seen in approximately 45% of CD30+ anaplastic large-cell lymphomas but is consistently absent in other peripheral T-cell lymphomas. Bio SB
Liquid amies collection and transport systems
Puritan liquid amies collection and transport systems are engineered to provide superior performance and ease of use in the collection and transport of clinically significant bacteria. Puritan’s proprietary HydraFlock swabs are designed with multi-length split fibers for greater sample collection capacity and faster elution. Puritan’s modified amies transport medium provides an ideal buffered environment to protect aerobic, anaerobic, and fastidious bacterial viability during collection and transport. Together, they comprise a system that increases the sensitivity of diagnostic tests. Puritan Medical Products Company
Determine HIV 1/2 Ag/Ab Combo
Alere has received U.S. Food and Drug Administration (FDA) approval of its pre-market application (PMA) to market Alere Determine(tm) HIV 1/2 Ag/Ab Combo in the United States. Alere Determine(tm) HIV-1/2 Ag/Ab Combo is the first and only FDA-approved rapid point-of-care test for the simultaneous and separate qualitative detection of HIV-1/2 antibodies and free H IV-1 p24 antigen, which can appear just days after infection and prior to HIV-1/2 antibodies. Alere
Whole blood lactate testing analyzer
StatStrip Lactate is a handheld analyzer for whole blood lactate testing. StatStrip Lactate’s single use POC biosensor provides accurate results in just 13 seconds on 0.6 µL of whole blood. Testing is performed at a fraction of the cost of lactate testing using a blood gas analyzer. Fast results, easy operation, and low cost make lactate testing practical in any department. Applications include routine screening of hospital patients for sepsis, serial lactate testing for goal-directed therapy of sepsis, septic shock, and other critical conditions. Nova Biomedical
Chemistry analyzer for the POL
The ACE Axcel is the third generation Chemistry analyzer from Alfa Wassermann. Designed to meet the needs of the physician office laboratory, it has been cleared by the FDA specifically for use in POLs. The ACE Axcel has touch screen functionality, an intuitive user interface, and Internet connectivity. A self-contained system with a small footprint, the Axcel can process up to 285 tests per hour. It can run any combination of single tests, panels or profiles, and has an open reagent system that enables custom assays. A stat interrupt feature allows users to load and prioritize stat samples during system operation to meet immediate diagnosis and treatment needs. Closed-tube sampling with the step module minimizes operator interaction with patient specimens, making for a safer workplace. Alfa Wassermann
Critical care analyzer
The GEM Premier 4000 now with plus technologycritical care analyzer measures pH, blood gases, electrolytes, metabolites, total bilirubin and co-oximetry (integrated). The analyzer features intelligent quality management(iQM)for continuous, real-time quality control, delivering the lab-quality results to any point-of-care location: ICU, NICU, CVOR, ED. Exceptionally easy to use, the GEM Premier 4000 is virtually maintenance-free and features the only single-component, multi-use cartridge on the market. GEMweb Plus custom connectivity allows remote control of all GEM Premier analyzers in the network for total connectivity, regardless of location, optimizing point-of-care flexibility. Plus technology offers faster touch screen response, wireless communication to HIS/LIS, and remote service capabilities. Instrumentation Laboratory
About the Author
Sign up for our eNewsletters
Get the latest news and updates