Sahara instruments automatically control the warming and thawing of blood products and I.V. fluids using dry heat. By providing an environment completely free of moisture, the Sahara instruments help to prevent potential growth of bacteria on the surface of the bags. If bacteria are allowed to grow, they may enter microscopic fractures in blood bags created during the freezing or storing process, causing contamination of products.
Three styles of Saharas are available, each with special features. The Sahara III has a warming plate for decreased thawing time of fresh frozen plasma; the Sahara Maxitherm has a double plate to increase capacity; the Sahara Thermal Box is an open warming unit with space for up to nine bottles of fluid. For added versatility, conversion modules for each model may be purchased to meet specific needs.
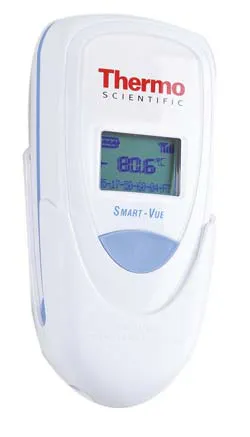
Don’t let a mechanical or electrical failure ruin your precious samples! The Smart-Vue wireless monitoring solution is a must-have for your ultra-low temperature (UTL) freezers, blood bank refrigerators, and plasma freezers. This innovative system safeguards the integrity of important laboratory storage by continuously monitoring critical parameters of laboratory equipment and securely logging data to ensure that sample integrity is upheld. Featuring audit trail traceability, the wireless monitoring system assists with conformance to 21 CFR part 11 and other regulatory requirements, making it ideal for use in highly controlled environments.
The system can be easily installed, set up, and maintained. It is compatible with multiple brands and types of laboratory equipment, including Thermo Scientific cold storage and centrifugation products for blood banking.
Transfusion management
Sunquest Laboratory™ Blood Bank integrates with Sunquest Transfusion Manager™ (TRM) as part of its closed loop protection of patient safety. Transfusion Manager’s FDA 510(k) clearance ensures that the nurse will not be able to proceed with a transfusion unless TRM has acknowledged that there is a match between the blood unit being administered and the patient undergoing the transfusion. TRM, therefore, as a 510(k) cleared medical device, can replace the second nurse verifier of the correct patient/blood cross-match prior to blood administration. Additionally, Transfusion Manager supports compliance with the Joint Commission National Patient Safety Goals by enabling nurses to carry out positive patient identification with two patient identifiers (NPSG 01.01.01), and eliminating transfusion errors related to patient identification (NPSG 01.03.01).
The PE-25-jw fully automated system has automatic process sequencing capabilities. It is equipped with PLC control and comes with a single gas channel, with the option of a second. All timed events (pumpdown, plasma, gas stabilization and vent) can be programmed into the automatic sequence. The PLC keypad is used for operator entry, and one complete process recipe can be store in memory for repeatable results. One button operation starts the process sequence. It has a direct RF contact (capacitive parallel plate) for uniform plasma generation. The unit has a 125W 50KHz RF power supply with continuously variable power capability. The unit has a 2.5″ x 4″ Horizontal “Direct Contact” RF powered electrode with 1.25″ of chamber height clearance.
Pevco TEC-6™ (Trackable Ergonomic Carrier, 6 inch) is designed with well placed hand-holds and contoured ends for efficient carrying and loading. Red/green latch status indicators help guarantee carriers are closed properly.
Integrated barcodes enable real-time tracking. When combined with Pevco Tracker™, transaction data and arrival confirmation for any carrier sent is stored in a database and available to authorized staff.
The carrier is constructed from BPA-free Eastman Tritan™ Copolyester, known for its super-durability, lasting clarity, and scratch resistance. It also features long-wearing Nylon glide bands, tough stainless steel hinges, high-density polyethylene end-caps developed to absorb landing impact, and leak-resistant seal.
It is designed to be used with 6″ hospital pneumatic tube delivery systems and is available in four color combinations. Fragile cargo can be transported with two options of foam inserts–pillow and egg crate.
An Anti-Dengue Mixed Titer Performance Panel has recently been launched by SeraCare as its first commercially available line of infectious disease panels responding to the demand for accurate testing for dengue virus. It is designed to help blood donor collection facilities, diagnostics manufacturers, and clinical laboratories evaluate and troubleshoot their anti-dengue virus assays. The new performance panel is derived from undiluted, unpreserved human plasma specimens, with 21 members representing a wide range of reactivity for anti-dengue IgM and IgG antibodies, from negative to strongly positive. Panel members include samples from individuals with recent and past infection by all four dengue viruses.
UltraCrit is a hand-held device that accurately measures hematocrit. It is the first and only hematocrit or hemoglobin device to use ultrasound technology.
Extensive tests have shown accuracy at <0.6% Hct (integer readout) and precision at <0.8% CV at 41 Hct. Results from fingerstick samples are highly correlated to the cell counter’s results on venous samples.
The device uses a single-use cuvette. A capillary blood sample is wicked into the cuvette, which is then inserted into the device. The measurement process takes about 30 seconds. No extensive training is required, and there is minimal chance of human error. The single-use cuvettes have a 15-month shelf life and contain no chemicals. The optional quality control material comes in single-use vials of low, normal and high levels and has a 30-month shelf life. It requires no refrigeration or mixing and is non-biohazardous. It can be run on wall power or 4-AA batteries and has a USB port.
The ELUclear kit provides a standardized glycine acid elution method that will dissociate clinically significant IgG antibodies bound to erythrocytes. The resulting eluate is used to identify the specificity using your laboratory's antibody identification procedure. ELUclear is the only eluate kit validated for use with Ortho™ MTS™ gel cards.
The kit combines clear end-point color change technology with convenience and versatility. Each kit contains color coded dropper vials and a wash bottle for ease of use and quality assurance.