Post-COVID-19: Long-term consequences with multiple manifestations
The COVID-19 pandemic was caused by the outbreak of severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2) in December 2019. While mild cases of COVID-19 may involve cold-like symptoms, severe cases can lead to hospitalization and/or death. Additionally, a new concern is developing, as long-term consequences of SARS-CoV-2 infection are being observed, even in patients whose disease course was mild or moderate.1,2 This phenomenon, referred to as long- or post-COVID-19, affects all age groups and is characterized by physical, cognitive, or psychological impairment.3 It is estimated that one in five individuals with a confirmed SARS-CoV-2 infection develop post-COVID-19 symptoms.4 These symptoms can last for a few weeks, months, or longer. Common post-COVID-19 symptoms include fatigue, shortness of breath, headache, anxiety, persistent cough, muscle pain, and difficulty thinking (brain fog). Additionally, risk factors have already been identified such as older age, cardiovascular disease, chronic lung disease, kidney disease, hypertension, and diabetes mellitus, initial disease severity, and female sex.2
According to the Centers for Disease Control and Prevention (CDC), at least four weeks after infection marks the start of when post-COVID-19 conditions can first be identified, as most people recover from the acute infection after a few weeks.5 Additionally, in 2021, the World Health Organization published the following working clinical case definition of post-COVID-19:
“Post COVID-19 condition occurs in individuals with a history of probable or confirmed SARS-CoV-2 infection, usually three months from the onset of COVID-19 with symptoms that last for at least two months and cannot be explained by an alternative diagnosis. Common symptoms include fatigue, shortness of breath, cognitive dysfunction but also others which generally have an impact on everyday functioning. Symptoms may be new onset, following initial recovery from an acute COVID-19 episode, or persist from the initial illness. Symptoms may also fluctuate or relapse over time. A separate definition may be applicable for children.”6
Several mechanisms have been proposed regarding post-COVID-19 conditions including immune dysregulation, microbiota dysbiosis, autoimmunity, blood clotting with endothelial abnormalities, and neurological signaling dysfunction.7 It has been proposed that autoimmune manifestations following COVID-19 are likely due to results of inflammatory cascade and the immune activation triggered by the virus, rather than a direct effect of the virus.8 Autoreactivity following SARS-CoV-2 might further be explained by a study that found that immunogenic peptides of SARS-CoV-2 have a high sequence homology with some human proteins.9
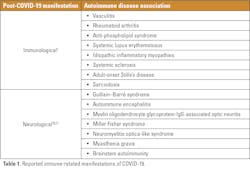
While a recent study found that many post-COVID-19 symptoms resolve on their own a year post infection, growing evidence that SARS-CoV-2 is associated with autoimmunity, suggests the possibility of long-term consequences.2,7,10 Such SARS-CoV-2 induced autoimmune disease might be a result of the production of disease-specific autoantibodies. This article will highlight the recent findings on the immunological and neurological manifestations of post-COVID-19 syndrome (Table 1).Immunological manifestations
Several studies have suggested that the formation of autoantibodies is involved in the development of post-COVID-19 syndrome.12,13 The presence of autoantibodies in patients with COVID-19 has been reported in different frequencies: antinuclear antibodies (ANA) in 50%, anti-Ro/SSA in 25%, rheumatoid factor in 19%, lupus anticoagulant in 11%.1,14,15 A meta-analysis study, that analyzed rheumatic autoimmune diseases in COVID-19 patients from December 2019 to September 2021, identified 99 patients that had fulfilled the diagnostic criteria for a specific rheumatic disease.1 The main diseases reported were vasculitis and arthritis, and a smaller number of patients were reported to have idiopathic inflammatory myopathies (IIM), systemic lupus erythematosus (SLE), sarcoidosis, systemic sclerosis, and adult-onset Still’s disease. Autoantibodies were reported in cases of IIM (anti-small ubiquitin-like modifier-1 activating enzyme, anti-Ku, anti-Mi 2b, anti-Ro/SSA, anti-Smith, anti-melanoma differentiation-associated protein 5) and SLE (anti-dsDNA antibodies, anti-Ro/SSA, anti-La/SSB, anti-histone, anti-RNP, anti-β2-glycoprotein I antibodies).1 Another study reported an increased prevalence of ANA, anti-neutrophil cytoplasmic antibodies (ANCA), and anti-Saccharomyces cerevisiae antibodies (ASCA) in 40 COVID-19 patients compared with healthy individuals. The authors proposed that autoimmunity is linked to SARS‐CoV‐2 infection, because none of the patients had a previous autoimmune disease.12
There have also been a number of studies reporting increases in anti-phospholipid antibodies in patients with COVID-19.10,16,17 Early in the pandemic, Zhang and colleagues found that patients with COVID‐19, with coagulopathy and multiple thrombi, were positive for anti‐cardiolipin IgA antibodies, anti-β2 glycoprotein 1 IgA antibodies, and IgG antibodies.18 Since then, the testing for anti-phospholipid antibodies in patients with COVID-19 has rapidly increased. Combined data from two studies depicted similar frequency of anti-phospholipid antibody positivity in COVID-19 patients admitted to intensive care units: 87% and 76% for lupus anticoagulant, 47% and 44% for anti-cardiolipin antibodies, 0% and 22% for anti-β2 glycoprotein I antibodies.10 However, several factors must also be considered when evaluating SARS-CoV-2 as a trigger of APS such as extent of association between anti-phospholipid antibodies and thrombosis, and persistence of antibody positivity, as positivity alone does not confirm APS.10
While there have been many reported cases of newly developed autoantibodies in COVID-19 patients, disease-specific autoantibodies have not been identified in every report of post-COVID-19 autoimmune disease. For example, anti-citrullinated protein antibodies (ACPA) have been found in cases of post-COVID-19 rheumatoid arthritis (RA); however, post-COVID-19 RA, without increased ACPA, has been reported also.19,20 Therefore, more research is still needed to understand the link between the autoantibodies and post-COVID-19 autoimmune diseases and to learn whether these “induced” antibodies differ from those normally occurring in RA and other autoimmune diseases.
The severity of SARS-CoV-2 infection might correlate with the specific immunological manifestation. Gracia-Ramos et al. found that more than half of the cases of post-COVID-19 arthritis (RA, spondyloarthritis and reactive arthritis) appeared in patients with mild COVID-19, vasculitis occurred in mostly mild or moderate cases, while more than half of IIM cases occurred in severe or critical COVID-19.1 Further, one study proposed that autoantibodies may also act to drive COVID-19 disease, as over 10% of patients with life-threatening pneumonia presented with antibodies against neutralizing interferon (IFN)-1, while patients with mild or asymptomatic COVID-19 infection had none.21 Therefore, pre-existence of anti-IFN-1 may be a risk factor for severe COVID-19 rather than a consequence of infection.
Neurological manifestations
Emerging research has shown a connection between SARS-CoV-2 and the nervous system.11 The most common neurological symptoms of COVID-19 are fatigue, concentration, memory disorders, headache, vertigo, myalgia and neuropathy, as well as persistent smell and taste disturbances.22 Additionally, post-COVID-19 neurological diseases have been described such as stroke, epileptic seizures, myelitis, Guillain-Barré syndrome (GBS), cranial nerve deficits, myositis, and plexopathies.11,22-24
Anti-neuronal autoantibodies have been found in cerebrospinal fluid (CSF) of severely ill COVID-19 patients suggesting an immune-mediated mechanism for post-COVID-19 neurological symptoms.25 In a 2021 study, serum and CSF samples were analyzed for anti-neuronal and anti-glial autoantibodies from critically ill COVID-19 patients presenting with unexplained neurological symptoms including myoclonus, oculomotor disturbance, delirium, dystonia, and epileptic seizures.25 Using cell-based assays and indirect immunofluorescence, anti-neuronal antibodies were detected in all samples. The autoantibodies detected were those against intracellular and neuronal surface proteins, such as Yo, myelin, and N-methyl-D-aspartate (NMDA) receptor. In another study, sera from patients with post-COVID-19 syndromes of neurological, cardiological origin, was found to contain functionally active autoantibodies targeting G-protein coupled receptors (GPCR). The specific targets of these autoantibodies included β2- and α1-adrenoceptors, angiotensin II AT1-, muscarinic M2-, MAS-, nociceptin-, and ETA-receptors. Future studies are needed to better understand the exact role of anti-GPCR antibodies in the development and maintenance of post-COVID symptoms. However, preliminary studies have begun to look at whether therapies targeting anti-GPCR antibodies could improve post-COVID-19 symptoms.26
A possible connection between COVID-19 and autoimmune encephalitis (AE) has been investigated. AE is a debilitating neurological disorder characterized by brain inflammation that leads to rapidly progressing encephalopathy. AE manifests with seizures and other neuropsychiatric symptoms. One study analyzed the frequency of SARS-CoV-2 antibodies in patients who underwent neural antibody testing as part of the diagnostic evaluation for AE at Mayo Clinic in 2020. This laboratory cohort was cross-referenced with the Department of Neurology's COVID-19–related consultative experience (encephalopathy cohort). Between both cohorts, a total of five patients were identified as having definite, probable, or possible AE representing 0.05% of all patients with COVID-19 illnesses evaluated. This, combined with other studies reporting anti-NMDA receptor antibodies post SARS-CoV-2 infection is evidence of a possible connection between COVID-19 and development of AE.
Several other autoimmune neurological disorders have been described in post-COVID-19 patients. Notably, a wide number of cases of peripheral nervous system involvement in the form of Guillain-Barré syndrome have been reported.23,24,27,28 In addition, multiple sclerosis has been reported in a patient following COVID-19 infection, and well as myelin oligodendrocyte glycoprotein antibody–associated optic neuritis and myelitis in another.29,30 Finally, antibodies against the brainstem proteins disabled homolog 1 (DAB1), apoptosis-inducing-factor-1 (AIFM1), and surfeit-locus-protein-1 (SURF1) have been found to be elevated in COVID-19 patients.31 Theses neuronal antigens are required for synaptic plasticity and higher cognition. While IgM levels were found to be comparable in both groups, IgG levels were significantly elevated in severely ill patients compared to controls, suggesting a pathogenic role of IgG.
Conclusion
Now over three years from the beginning of COVID-19 pandemic, there have been many reported cases of autoimmunity following SARS-CoV-2 infection. The autoimmune manifestations of post-COVID-19 syndrome include rheumatic, neurologic, dermatologic, and cardiac disorders. There are no current testing guidelines for long- or post-COVID-19 in the United States; however, guidelines have been developed in other countries, such as Germany.32 While there is no one test for post-COVID-19 syndrome, as this condition is not a single illness, there are assays available for the detection of specific autoantibodies that are increased in post-COVID-19 patients.
More research is still needed to better understand the link between SARS-CoV-2 and new onset-autoimmune diseases. Questions that remain include: Are SARS-CoV-2 induced antibodies different from the typical disease-associated antibodies? Are post-COVID-19 autoimmune manifestations transitory or might they persist longer resulting in chronic conditions? What is the exact mechanism of SARS-CoV-2–influenced autoantibody development? This additional knowledge could then be applied to achieve the optimal testing strategy and determine the most suitable therapy.
REFERENCES
- Gracia-Ramos AE, Martin-Nares E, Hernández-Molina G. New Onset of Autoimmune Diseases Following COVID-19 Diagnosis. Cells. 2021;20;10(12):3592. doi:10.3390/cells10123592.
- Mizrahi B, Sudry T, Flaks-Manov N, Yehezkelli Y, et al. Long covid outcomes at one year after mild SARS-CoV-2 infection: nationwide cohort study. BMJ. 2023;11;380:e072529. doi:10.1136/bmj-2022-072529.
- Lopez-Leon S, Wegman-Ostrosky T, Perelman C, Sepulveda R, et al. More than 50 long-term effects of COVID-19: a systematic review and meta-analysis. Sci Rep. 2021;9;11(1):16144. doi:10.1038/s41598-021-95565-8.
- Long COVID. Cdc.gov. Published January 24, 2023. Accessed February 20, 2023. https://www.cdc.gov/nchs/covid19/pulse/long-covid.htm.
- CDC. Long COVID or post-COVID conditions. Centers for Disease Control and Prevention. Published December 19, 2022. Accessed February 20, 2023. https://www.cdc.gov/coronavirus/2019-ncov/long-term-effects/index.html.
- A clinical case definition of post COVID-19 condition by a Delphi consensus, 6 October 2021. Who.int. Published October 6, 2021. Accessed February 20, 2023. https://www.who.int/publications/i/item/WHO-2019-nCoV-Post_COVID-19_condition-Clinical_case_definition-2021.1.
- Davis HE, McCorkell L, Vogel JM, Topol EJ. Long COVID: major findings, mechanisms and recommendations. Nat Rev Microbiol. 2023;21(3):133-146. doi:10.1038/s41579-022-00846-2.
- Novelli L, Motta F, De Santis M, Ansari AA, Gershwin ME, Selmi C. The JANUS of chronic inflammatory and autoimmune diseases onset during COVID-19 - A systematic review of the literature. J Autoimmun. 2021;117:102592. doi:10.1016/j.jaut.2020.102592.
- Lyons-Weiler J. Pathogenic priming likely contributes to serious and critical illness and mortality in COVID-19 via autoimmunity. J Transl Autoimmun. 2020;9;3:100051. doi:10.1016/j.jtauto.2020.100051.
- Ramos-Casals M, Brito-Zerón P, Mariette X. Systemic and organ-specific immune-related manifestations of COVID-19. Nat Rev Rheumatol. 2021;17(6):315-332. doi:10.1038/s41584-021-00608-z.
- Ellul MA, Benjamin L, Singh B, Lant S, et al. Neurological associations of COVID-19. Lancet Neurol. 2020;19(9):767-783. doi:10.1016/S1474-4422(20)30221-0.
- Sacchi MC, Tamiazzo S, Stobbione P, Agatea L, et al. SARS-CoV-2 infection as a trigger of autoimmune response. Clin Transl Sci. 2021;14(3):898-907. doi:10.1111/cts.12953.
- Vlachoyiannopoulos PG, Magira E, Alexopoulos H, Jahaj E, et al. Autoantibodies related to systemic autoimmune rheumatic diseases in severely ill patients with COVID-19. Ann Rheum Dis. 2020;79(12):1661-1663. doi:10.1136/annrheumdis-2020-218009.
- Chang SE, Feng A, Meng W, Apostolidis SA, et al. New-onset IgG autoantibodies in hospitalized patients with COVID-19. Nat Commun. 2021;14;12(1):5417. doi:10.1038/s41467-021-25509-3.
- Zhou Y, Han T, Chen J, Hou C, et al. Clinical and Autoimmune Characteristics of Severe and Critical Cases of COVID-19. Clin Transl Sci. 2020;13(6):1077-1086. doi:10.1111/cts.12805.
- Garcia-Arellano G, Camacho-Ortiz A, Moreno-Arquieta IA, Cardenas-de la Garza JA, et al. Anticardiolipin and anti-beta-2 glycoprotein I antibodies in patients with moderate or severe COVID-19. Am J Med Sci. 2023;365(2):215-217. doi:10.1016/j.amjms.2022.10.012.
- Serrano M, Espinosa G, Serrano A, Cervera R. COVID-19 and the antiphospholipid syndrome. Autoimmun Rev. 2022;21(12):103206. doi:10.1016/j.autrev.2022.103206.
- Zhang Y, Xiao M, Zhang S, Xia P, et al. Coagulopathy and Antiphospholipid Antibodies in Patients with Covid-19. N Engl J Med. 2020;23;382(17):e38. doi:10.1056/NEJMc2007575.
- Perrot L, Hemon M, Busnel JM, Muis-Pistor O, et al. First flare of ACPA-positive rheumatoid arthritis after SARS-CoV-2 infection. Lancet Rheumatol. 2021;3(1):e6-e8. doi:10.1016/S2665-9913(20)30396-9.
- Gasparotto M, Framba V, Piovella C, Doria A, Iaccarino L. Post-COVID-19 arthritis: a case report and literature review. Clin Rheumatol. 2021;40(8):3357-3362. doi:10.1007/s10067-020-05550-1.
- Bastard P, Rosen LB, Zhang Q, Michailidis E, et al. Autoantibodies against type I IFNs in patients with life-threatening COVID-19. Science. 2020;23;370(6515):eabd4585. doi:10.1126/science.abd4585.
- Franke C, Berlit P, Prüss H. Neurological manifestations of post-COVID-19 syndrome S1-guideline of the German society of neurology. Neurol Res Pract. 2022;18;4(1):28. doi:10.1186/s42466-022-00191-y.
- Abu-Rumeileh S, Abdelhak A, Foschi M, Tumani H, Otto M. Guillain-Barré syndrome spectrum associated with COVID-19: an up-to-date systematic review of 73 cases. J Neurol. 2021;268(4):1133-1170. doi:10.1007/s00415-020-10124-x.
- Kaeley N, Kabi A, Pillai A, Shankar T, Ameena M S S. Post-COVID-19 Guillain-Barré Syndrome: A Case Report With Literature Review. Cureus. 2022;14;14(1):e21246. doi:10.7759/cureus.21246.
- Franke C, Ferse C, Kreye J, Reincke SM, et al. High frequency of cerebrospinal fluid autoantibodies in COVID-19 patients with neurological symptoms. Brain Behav Immun. 2021;93:415-419. doi:10.1016/j.bbi.2020.12.022.
- Hohberger B, Harrer T, Mardin C, Kruse F, et al. Case Report: Neutralization of Autoantibodies Targeting G-Protein-Coupled Receptors Improves Capillary Impairment and Fatigue Symptoms After COVID-19 Infection. Front Med (Lausanne). 2021;18;8:754667. doi:10.3389/fmed.2021.754667.
- Alberti P, Beretta S, Piatti M, Karantzoulis A, et al. Guillain-Barré syndrome related to COVID-19 infection. Neurol Neuroimmunol Neuroinflamm. 2020;29;7(4):e741. doi:10.1212/NXI.0000000000000741.
- Zhao H, Shen D, Zhou H, Liu J, Chen S. Guillain-Barré syndrome associated with SARS-CoV-2 infection: causality or coincidence? Lancet Neurol. 2020;19(5):383-384. doi:10.1016/S1474-4422(20)30109-5.
- Palao M, Fernández-Díaz E, Gracia-Gil J, Romero-Sánchez CM, Díaz-Maroto I, Segura T. Multiple sclerosis following SARS-CoV-2 infection. Mult Scler Relat Disord. 2020;45:102377. doi:10.1016/j.msard.2020.102377.
- Palao M, Fernández-Díaz E, Gracia-Gil J, Romero-Sánchez CM, Díaz-Maroto I, Segura T. Multiple sclerosis following SARS-CoV-2 infection. Mult Scler Relat Disord. 2020;45:102377. doi:10.1016/j.msard.2020.102377.
- Lucchese G, Vogelgesang A, Boesl F, Raafat D, et al. Anti-neuronal antibodies against brainstem antigens are associated with COVID-19. EBioMedicine. 2022;83:104211. doi:10.1016/j.ebiom.2022.104211.
- Rabady S, Altenberger J, Brose M, Denk-Linnert DM, et al. Leitlinie S1: Long COVID: Differenzialdiagnostik und Behandlungsstrategien [Guideline S1: Long COVID: Diagnostics and treatment strategies]. Wien Klin Wochenschr. 2021;133(Suppl 7):237-278. German. doi:10.1007/s00508-021-01974-0.
About the Author

Ilana Heckler, PhD
is the Scientific Affairs Liaison at EUROIMMUN US. In this role, Heckler supports scientific collaborations and assists in the validation of diagnostic assays for autoimmune and infectious diseases.