The clinical laboratory’s role in diagnosis and management of hypopituitarism
Earning CEUs:
For a printable version of the November CE test go HERE or to take test online go HERE. For more information, visit the Continuing Education tab.
NOVEMBER LEARNING OBJECTIVES
Upon completion of this article, the reader will be able to:
1. Recall the anatomy of the pituitary gland and the hormones originating from it.
2. Describe hypopituitarism, its causes and etiology.
3. Discuss diagnostic tests for a variety of hormone disorders.
4. Describe patient and technical considerations when testing for hormone disorders.
The clinical laboratory is central to the diagnosis and management of many endocrine disorders. Tests availability, as well as their performance characteristics, must allow for both timely and accurate information. This report, inspired by recent clinical practice guidelines,1 describes the clinical laboratory’s role in the diagnosis and management of patients with hypopituitarism, an often-underestimated endocrine disorder.
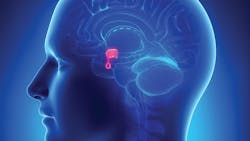
The pituitary
The pituitary gland occupies the pituitary Sella. It is divided into two anatomically and functionally distinct structures known as the anterior and posterior segments. The anterior pituitary segment is made up of five cell types. The lactotrophs synthesizing prolactin, the somatotrophs synthesizing growth hormone (GH), the corticotrophs synthesizing adrenocorticotropic hormone (ACTH) and Melanocyte stimulating hormone (MSH), the thyrotrophs synthesizing thyroid-stimulating hormone (TSH), and the gonadotrophs synthesizing luteinizing (LH) and follicle-stimulating (FSH) hormones. The posterior pituitary is innervated by nerve cells originating in the hypothalamus. It mainly acts as reservoir for release of hypothalamic hormone’s namely anti-diuretic hormone (ADH) and oxytocin. Dysfunction of the pituitary manifests in either hyper or hypofunction of the gland with corresponding changes in circulating hormonal levels and clinical presentation.
Hypopituitary
Evident by either complete or partial deficiency of pituitary hormones, the etiology of hypopituitarism is variable and could be due to either disorders causing hormonal release dysfunction of the pituitary tissue or secondary to dysfunctional hypothalamic release of pituitary hormones releasing hormones.
Although probably underestimated, prevalence of the condition is 45 cases per 100,000 with an incidence of four cases per 100,000 per year.2 Although the incidence and prevalence are variable among populations,2,3 a common trend of increase in both is apparent. The observed increase in mortality is attributed to ensued cardiovascular and respiratory disease. Interestingly, mortality is higher among women compared to men,4,5 suggesting a role of sex hormones on outcomes.
Congenital or acquired hypopituitarism
Causes of hypopituitarism can be classified as either congenital or acquired. In acquired hypopituitarism, pituitary adenoma, pituitary surgery and radiotherapy are the major causes in adults. Primary or secondary pituitary tumors cause pituitary tissue loss and thus, function via compression of the portal vessels (leading to ischemia). Similarly, physical space occupying lesions and large tumors cause increased intrasellar pressure and pituitary tissue ischemia (Sheehan’s syndrome).
Pituitary surgery is the second most common cause and although newer targeted radiation therapy is being used, hypopituitarism can still occur as a consequence. Traumatic brain injury (TBI) causes hypopituitary. Similarly, pituitary apoplexy due to infarction or hemorrhage causes a rapid hormonal deficiency. Physiological increase in pituitary volume during normal pregnancy, mainly due to hyperplasia of the lactotrophs, may exceed blood flow requirements leading to postpartum hemorrhage, ischemia and infarction.6
Sarcoidosis, tuberculosis and other granulomatous disease can cause hypopituitarism. Iron deposition in conditions of iron overload, such as haemochromatosis, causes hypopituitarism. Diabetes insipidus is more common in patients with infiltrative disorders, whereas hypogonadism is more common in patients with iron overload.
Immune-mediated infiltration of lymphocytes and plasma cells causes hypopituitarism mainly affecting the anterior pituitary. Inflammation of the pituitary is seen in patients undergoing immunotherapy with certain humanized monoclonal antibody therapy.7
The pituitary has a large reserve capacity, and the deficiency is often realized following infection, stress or an insult. Which hormones are affected depend on the pituitary cells affected and the underlying pathology. Some are life-threatening, requiring immediate intervention such as adrenal crisis, while others present over a longer time period such as infertility (pituitary apoplexy).
Congenital hypopituitarism
Congenital hypopituitarism arises due to mutations in one or several of the genes involved in the development and functional activity of the various pituitary tissue. Genetic lineage varies from being autosomal recessive, and/or autosomal dominant, as well as x-linked mutations. It can be isolated deficiency as for growth hormone or combined pituitary hormones deficiency. There are also rare and poorly understood mutations causing isolated TSH, ACT, FSH and LH deficiencies. Identification based on clinical suspicion begins around age five years for GH deficiency, followed by recognition for TSH and gonadotrophins.8
Mutation in the PROP1, PROP1F1, LHX3 and LHX4 genes is associated with complete pituitary hormones deficiency whereas, isolated growth hormone deficiency are associated with GH1 genes mutations, 1-A, 1-B, and 2.9
Geographical distribution for the causes of hypopituitarism have been identified. For instance, in developed countries, pituitary tumors and radiation therapy are the most common causes of hypopituitarism, whereas developing countries and the tropics are often due to inflammatory and infective causes. Similarly, Sheehan’s syndrome is more frequent in areas with less developed obstetric care.
A recent Endocrine Society Guideline addressing the diagnosis and management of patients has been published.1 Clinical practice guidelines were prepared using the Grading of Recommendations, Assessment, Development and Evaluation (GRADE) approach.10
Diagnostic tests
Clinical laboratory tests are central to both diagnosis and management. However, the diagnosis of hypopituitarism can be difficult, and evidence suggests it is often missed with significant morbidities. In addition to clinical diagnosis, laboratory measurements of pituitary hormones are central to confirming and establishing diagnosis of hypopituitarism, and of monitoring replacement therapy. Understanding hormonal assay characteristics and limitations are thus important. Technical considerations include sample stability as assays are often added-on a few hours or even days following presentation, and assay characteristics such as sensitivity, interference from metabolites, and from replacement hormones and their analogous.
Patients often present with symptoms due to secondary organ failure, such as adrenal insufficiency due to deficient pituitary ACTH or hypothyroidism due to deficient pituitary TSH. Patients suspected of adrenal insufficiency secondary to hypopituitary is confirmed by measuring serum cortisol at 8—9 AM as the first line tests for the diagnosis of central adrenal insufficiency where a level <3ug/dL confirms suspicion and value >15ug/dL excludes adrenal insufficiency.
Synthetic glucocorticoids, such as prednisolone, prednisone and 6-alpha methylprednisolone, may interfere with cortisol immunoassays. Where patients may be on replacement therapy, the guidelines recommend assessment for hypothalamic-pituitary-adrenal (HPA) axis 18 to 24 hours following cessation of hydrocortisone or longer for synthetic glucocorticoids. It is recommended that samples be collected before initiation of therapy for prednisolone, however, dexamethasone exhibits very low or no reactivity in most immunoassays.
Patients may present with secondary hypothyroidism. In those patients, both thyroidal FT4 and pituitary TSH will be low or at the lower end of the reference range confirming the clinical diagnosis. A decrease in FT4 by more than 20 percent confirms the diagnosis in those without clinical symptoms.
In patients suspected of GH deficiency, whereas a single random measurement is not helpful, use of body mass index (BMI) adjusted GH peak levels is recommended.
In patients suspected of secondary hypogonadism, measurement of FSH and LH, as well as prolactin either before 10 AM (after an overnight fast), confirms the diagnosis in males. However, in females, estradiol, as well as hCG (pregnancy test), are added to the test menu. In postmenopausal women, expect to see elevated FSH and low levels of LH, which suggests hypopituitarism. However, dynamic tests of function by hyperstimulation are no longer recommended for TSH secretion, for LH/FSH (GnRH).
In patients suspected of diabetes insipidus, the finding of polyuria (defined as >50mL/Kg body weight/24hours) in the absence of glucosuria (determined by dip stick) and the finding of a high serum osmolality (>295 mOsmol/L) and inappropriately low urine osmolarity (<600 mOsmol/L) indicates diabetes insipidus.
Patient and technical considerations
Patient preparation (prandial diurnal status), medication, as well as sample type and sample transportation and preservation, are important considerations when measuring and interpreting hormonal results.
Replacement therapy (substitution) of the deficient hormone is the cornerstone of patient management. Although not often practical, replacement should be tailored to resemble physiological patterns such as pulsatility and diurnal rhythm. However, replacement therapy is complicated by coexistence of disorders, including patients on anti-epileptic drugs and other medication affecting metabolism, stress and surgery, as well as pregnancy.
Anti-epileptic drugs increase hormone metabolism by stimulating hepatic microsomal enzymes, CYP450.11 This hastens their metabolism and thus, decreases their half-life and circulating concentrations. The extent of metabolism depends on the CYP450 isoenzyme and on the particular drug—for example, CYP3A4 preferentially metabolizes dexamethasone. The above effect of antileptics is also seen for patients on T4 and estradiol, as well as DDAVP. Increased metabolism and degradation of therapeutic glucocorticoids places the patient at increased risk for adrenal insufficiency and adrenal crisis. The guidelines recommend dosage adjustment for those patients to compensate for the increased metabolism. Monitoring the levels of those supplemental hormones is essential in guiding dosage adjustment.
Furthermore, anti-epileptics are also known to displace hormones from their binding proteins leading to falsely elevated free hormones concentrations. Questionable FT4 should be analyzed by equilibrium dialysis. The drugs also increase sex hormones binding globulin (SHBG) levels, causing a reduction in bioavailable estradiol (E2) and testosterone. Interestingly, combined use of carbamazepine, oxcarbazepine, lamotrigine, perampanle or felbamate increase renal tissue responsiveness to DDAVP.11
Commercially available assays for hormone measurements, and those particularly in wide use, have acceptable sensitivities for the purpose of investigation of low hormone concentrations seen in hypopituitarism. Although intraassay imprecision was acceptable, interassays were problematic for estradiol, testosterone, GH, FT4 and gonadotrophins. When performing serial measurements, the use of the same assay is recommended.1
Interference in ACTH assays due to either presence of heterophile antibodies or ACTH fragments occur. Falsely elevated levels, which what is often seen, leads to missing hypopituitary adrenal insufficiency and may lead to unnecessary delay and investigation and even surgical exploration.12 ACTH precursors pro-opiomelanocortin (POMC) and pro-ACTH have been identified in circulation in normal subjects, which may interfere in some non-specific ATCH assays giving misleading elevated or normal results.13 Those may interfere with non-specific ACTH immunoassays with several reports on interference due to ectopic (non-pituitary) sources of ACTH, thought to have incomplete ACTH synthesis and those with POMC secretion.14 Additionally, negative interference in ACTH assays is seen in patients on ACTH 1-24 therapy.15
The issue of heterophile antibodies interfering with hormonal assays, although widely known, is not often tested for until several test repeats, medical and surgical interventions, delayed treatment, or unexpected outcomes had occurred. Although commercially available heterophile antibodies blocking reagents are helpful, they may not work for some.16
Recent reports describe interferences in avidin-biotin based assays, where excessive intake of biotin leads to false hormonal results.17 Cases of false biochemical hyperthyroidism (suppressed TSH and elevated FT4) have been reported.18 Circulating biotin has a half-life between 8—18 hours, and a repeat specimen eight hours following cessation of intake, resolves the discrepancy.
In support of endocrine testing
Therefore, in support of endocrine testing, the clinical laboratory needs to be conversant with assay performance characteristics. In general, significant variability among assay reportable values were observed. Those are due to use of different calibrators (standards), and lack of standardization (use of a consensus international reference preparations (IRP)), and lack of commutability). Assay sensitivities were appropriate when compared against published diagnostic protocols. Sample stabilities were overall appropriate with the exception of a few with variable recommendations on storage.
Increasing use of liquid chromatography tandem mass-spectrometry (LC-MS-MS) has afforded many advantages for hypopituitary patients. For instance, it affords the required sensitivity where hormonal levels are expected to be low and the required specificity such as distinguishing between cortisol and synthetic glucocorticoids. It is however, important to note that reports of inferences with LC-MS-MS methods exist such as interference form gel-separator material containing compounds with similar characteristics to steroids testosterone.19
LC-MS-MS methods allow multiple analytes measurements on a single sample, including those replacement therapies where reliance on FT4 measurement in patients with hypopituitarism requires measurement by LCMS/MS.20 The methodology, however requires technical expertise and is not amenable to the rapid turnaround time often required in patients with hypopituitary emergencies.
Studies have shown that LC-MS-MS is superior to immunoassays when measuring thyroid hormones. The FT4 hormones correlate better with logarithmic TSH concertation when compared with other immunoassays. Additionally, patients with binding proteins abnormalities (albumin and SHBG) and those with non-thyroidal illness remain problematic.
In conclusion
Diagnosis and management of hypopituitarism requires clinical laboratory support. Currently available laboratory methodologies, although usable for most, suffer from significant interferences. The clinical laboratory is responsible for educating the clinicians on the utility of those assays.
REFERENCES
- Fleseriu M, Hashim IA, Karavitaki N, et al. Hormonal Replacement in Hypopituitarism in Adults: An Endocrine Socity Clinical Practice Guideline. J Clin Endocrinol Metab 2016; 101(11):3888-921 doi: 10.1210/jc.2016-2118 [Published Online First: Epub Date].
- Regal M, Paramo C, Sierra SM, Garcia-Mayor RV. Prevalence and incidence of hypopituitarism in an adult Caucasian population in northwestern Spain. Clin Endocrinol (Oxf) 2001; 55(6):735-40.
- Nilsson B, Gustavasson-Kadaka E, Bengtsson BA, Jonsson B. Pituitary adenomas in Sweden between 1958 and 1991: Incidence, survival, and mortality. J Clin Endocrinol Metab 2000;85(4):1420-5 doi: 10.1210/jcem.85.4.6498[published Online First: Epub Date].
- Tomlinson JW, Holden N, Hills RK, et al. Association between premature mortality and hypopituitarism. West Midlands Prospective Hypopituitary Study Group. Lancet 2001;357(9254):425-31 doi: 10.1016/s0140-6736(00)04006-x[published Online First: Epub Date].
- Sherlock M, Ayuk J, Tomlinson JW, et al. Mortality in patients with pituitary disease. Endocr Rev 2010;31(3):301-42 doi: 10.1210/er.2009-0033[published Online First: Epub Date].
- Diri H, Tanriverdi F, Karaca Z, et al. Extensive investigation of 114 patients with Sheehan’s syndrome: a continuing disorder. Eur J Endocrinol 2014;171(3):311-8 doi: 10.1530/EJE-14-0244[published Online First: Epub Date].
- Faje AT, Sullivan R, Lawrence D, et al. Ipilimumab-induced hypophysitis: a detailed longitudinal analysis in a large cohort of patients with metastatic melanoma. J Clin Endocrinol Metab 2014;99(11):4078-85 doi: 10.1210/jc.2014-2306[published Online First: Epub Date].
- Mody S, Brown MR, Parks JS. The spectrum of hypopituitarism caused by PROP1 mutations. Best Pract Res Clin Endocrinol Metab 2002;16(3):421-31.
- Parks JS. Congenital Hypopituitarism. Clin Perinatol 2018;45(1):75-91 doi: 10.1016/j.clp.2017.11.001[published Online First: Epub Date].
- Swiglo BA, Murad MH, Schunemann HJ, et al. A case for clarity, consistency, and helpfulness: state-of-the-art clinical practice guidelines in endocrinology using the grading of recommendations, assessment, development, and evaluation system. J Clin Endocrinol Metab 2008;93(3):666-73 doi: 10.1210/jc.2007-1907[published Online First: Epub Date].
- Paragliola RM, Prete A, Kaplan PW, Corsello SM, Salvatori R. Treatment of hypopituitarism in patients receiving antiepileptic drugs. Lancet Diabetes Endocrinol 2015;3(2):132-40 doi: 10.1016/S2213-8587(14)70081-6[published Online First: Epub Date].
- Greene LW, Geer EB, Page-Wilson G, Findling JW, Raff H. Assay-Specific Spurious ACTH Results Lead to Misdiagnosis, Unnecessary Testing, and Surgical Misadventure-A Case Series. J Endocr Soc 2019;3(4):763-72 doi: 10.1210/js.2019-00027[published Online First: Epub Date].
- Oliver RL, Davis JR, White A. Characterisation of ACTH related peptides in ectopic Cushing’s syndrome. Pituitary 2003;6(3):119-26.
- Page-Wilson G, Freda PU, Jacobs TP, et al. Clinical utility of plasma POMC and AgRP measurements in the differential diagnosis of ACTH-dependent Cushing’s syndrome. J Clin Endocrinol Metab 2014;99(10):E1838-45 doi: 10.1210/jc.2014-1448[published Online First: Epub Date].
- Raff H, Findling JW, Wong J. Short loop adrenocorticotropin (ACTH) feedback after ACTH-(1-24) injection in man is an artifact of the immunoradiometric assay. J Clin Endocrinol Metab 1989;69(3):678-80 doi: 10.1210/jcem-69-3-678[published Online First: Epub Date].
- Grasko J, Willliams R, Beilin J, Glendenning P, Fermoyle S, Vasikaran S. A diagnostic conundrum: heterophilic antibody interference in an adrenocorticotropic hormone immunoassay not detectable using a proprietary heterophile blocking reagent. Ann Clin Biochem 2013;50(Pt 5):433-7 doi: 10.1177/0004563213487514[published Online First: Epub Date].
- Holmes EW, Samarasinghe S, Emanuele MA, Meah F. Biotin Interference in Clinical Immunoassays: A Cause for Concern. Arch Pathol Lab Med 2017;141(11):1459-60 doi: 10.5858/arpa.2017-0107-LE[published Online First: Epub Date].
- Piketty ML, Polak M, Flechtner I, Gonzales-Briceno L, Souberbielle JC. False biochemical diagnosis of hyperthyroidism in streptavidin-biotin-based immunoassays: the problem of biotin intake and related interferences. Clin Chem Lab Med 2017;55(6):780-88 doi: 10.1515/cclm-2016-0606[published Online First: Epub Date].
- Shi RZ, van Rossum HH, Bowen RA. Serum testosterone quantitation by liquid chromatography-tandem mass spectrometry: interference from blood collection tubes. Clin Biochem 2012;45(18):1706-9 doi: 10.1016/j.clinbiochem.2012.08.008[published Online First: Epub Date]
- van Deventer HE, Soldin SJ. The expanding role of tandem mass spectrometry in optimizing diagnosis and treatment of thyroid disease. Adv Clin Chem 2013;61:127-52
About the Author

Ibrahim A. Hashim, MSc, PhD
serves as Professor of Pathology, Chief of Clinical Pathology and Clinical Chemistry Director at the University of Texas Southwestern Medical Center, Dallas, TX. A graduate of King’s College London, UK, with interest in pituitary, neuroendocrine biomarkers, and the acute phase response. Hashim is a co-author of the Endocrine Society’s Hormone Replacement in Hypopituitarism in Adults Clinical Practice Guideline.