Middleware systems have become a “must have” not only for large laboratories, but, increasingly, for physician office laboratories (POLs) and clinics, providing these labs with instrument connectivity directly to the electronic medical record (EMR). Middleware is the primary solution to automate processes and workflow in order to reduce labor utilization and improve the quality of lab reporting. The convergence of data availability and ready-made reporting tools provides the laboratory, large or small, with the ability to monitor its operation. Middleware reporting tools are like a “GPS” for the laboratory; they get you where you want to go. Middleware assists the laboratory in determining whether operations are on the right track. It helps lab managers answer the questions: Are we moving in the right direction? Where do we need to go next to increase efficiency?
Beyond auto-validation
Regardless of size or workload, auto-validation rates have become one of the indicators by which the success of a middleware system can be evaluated. With the appropriate set of decision-based rules that also consider the patient population, validation rates of >85 percent have been achieved. Using middleware with business reporting tools can help highlight areas for improvement by:
- helping to focus on what is important for the laboratory to monitor
- substantiating assumptions about performance
- benchmarking data over time and across patient acuity
- providing the ability to adapt faster once trends are detected.
The availability of reporting tools helps a laboratory increase its acceleration once trends are identified, and also enables lab leaders to anticipate the impact of environmental changes once the data is better understood. A lab can react faster to optimize its operation and build on the complex relationship between clinical and operational data; an insight into the linkage can highlight changes that need to be made in optimizing operational practices and staffing activities. Data is a few clicks away.
Count statistics reporting
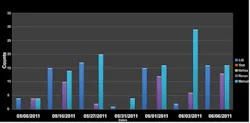
Count Statistics Reports tally the number of tests and provide ratios for a look at laboratory productivity for rerun, reflex, stat, and auto-validation to manual validation rates. These reports can drill down and determine the auto-validation rates by site, location, and provider and/or across the enterprise. This type of data provides the laboratory the ability to benchmark its rates by its own patient mix and acuity. For example, managers can identify their auto-validation rates by hour, day and/or shift to measure and monitor productivity down to the user level. This type of data can help lab managers and supervisors answer key questions such as, “What resources need to be moved or changed to meet increase/decrease in demand?” Data like this can help the laboratory anticipate environmental changes quicker and make the adjustment faster (Figure 1).
TAT statistics reporting
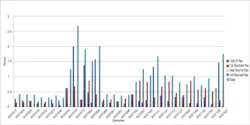
Turnaround Time Statistics Reports are used to obtain information about laboratory productivity and reporting. Middleware tools usually can provide information on discrete time points in regard to the laboratory information system (LIS) receipt times, instrumentation receipt, first availability of data, and last review of data before the release to the LIS. The Turnaround Time Statistics Report provides more discrete in-depth tracking of possible gaps in handling of the specimen and results to the LIS. The key benefit of monitoring and benchmarking the lab turnaround time by profile or test is the identification of reporting delays along the workflow path (Figure 2).
Rules statistics reporting
The Rules Statistics Report further assesses the health and utilization of the auto-validation rules. It helps monitor rule utilization by rule type. This report is designed to observe rule trending such as increases in rule usage by a specific instrument or patient location or provider, thus helping the lab to dig deeper to determine the reason for the change.
Are too many smears being made? Does this reflect a lower than expected auto-validation rate? Is there a higher than expected manual review rate?
The use of the Rule Statistics Report, in conjunction with the auto-validation rate, is a key metric in assisting the laboratory in fine tuning rule usage and maintaining the health of the auto-validation rule base.
Results statistics reporting
The Result Statistics Report can be used as a data mining report to help correlate and cross-verify data trends. It can help dig deeper beyond the surface to answer a specific question and/or identify data relationships that were not expected. The typical use of this type of report is to compare data between methods, such as how many hemoglobin values were critically low, whether the results were called in to a clinician, and how these findings correlate with product transfusion rates by the organization. The Result Statistics Report can help identify organizational issues:
Are there too many delta failures from a specific location? Can this be correlated with misdraws or misidentified patients? What workflow changes can be made across the organization to eliminate patient redraw rates?
This type of report can help make sense of the lab environment; the power of combing and cross-correlating clinical data can help assist in making this connection.
Like other analytic technology improvements across laboratory disciplines, middleware has been designed for a variety of clinical settings. From the physician office laboratory to a multi-site, multi-location enterprise, connectivity and analytical prowess have been brought together, not only as mechanisms for improving laboratory performance, but for consolidating and reporting of disparate information to clinicians. Middleware is data-rich and offers the most efficient tool to retrieve a report on demand that could take months from other sources. Data reporting tools give the laboratory the means to identify trends and to act faster to reduce errors and improve efficiency. They put the laboratory in the driver’s seat to fine-tune its environment and to stay competitive while helping to enhance the quality of patient care.