Understanding sepsis: The importance of biomarkers in early diagnosis
It starts with a common infection leading to a chain reaction and deterioration of a person’s health. The symptoms are aggressive — including an accelerated heart rate, fever, shivering, confusion or disorientation, shortness of breath, and extreme pain. It’s common to hear patients say, “It feels like I am going to die.” When an infection persists or gets worse, and it could lead to sepsis and septic shock.
Sepsis, a life-threatening organ dysfunction in response to infection, affects 47–50 million people worldwide each year with approximately 11 million deaths.1 With an overall mortality rate of 15–30% leading to 30–50% of all in-hospital mortality.2 Sepsis is the most expensive condition in modern medicine. The worldwide incidence of sepsis continues to increase, putting a high financial burden on society and the healthcare system.2 More individuals die of sepsis than prostate cancer, breast cancer, and HIV combined.3,4 Because anyone can get an infection, anyone can develop sepsis.
There are several factors contributing to the high toll of sepsis. When it comes to sepsis treatment, time to source control matters. For every hour treatment is delayed, there is up to a 7.9% increase in mortality and as much as a 10% increase in the odds of one-year mortality.5,6 Early and accurate identification of sepsis is needed for successful treatment and a positive outcome.5
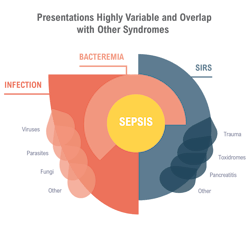
Most cases of sepsis are diagnosed in the emergency department (ED) with 87% of cases starting prior to admission.7 When patients arrive at the ED, clinicians are tasked with quickly triaging patients based on limited diagnostic information. As the patient struggles to understand what is happening, the ED clinicians face the challenging task of assessing and determining a course of action. Sepsis identification relies on a combination of clinical suspicions based on nebulous patient symptoms with laboratory test results that can indicate multiple conditions (see Figure 1). Unfortunately, failure to recognize sepsis early in the disease course often leads to worse outcomes.8
Sepsis diagnosis requires confirmation of an underlying infection through a positive blood culture, but these tests can take two to three days for results9 — too long for defining a sepsis care pathway. Moreover, the overlap of clinical symptoms between sepsis and other, non-infectious inflammatory conditions can confound the diagnosis.
The challenge is to diagnose sepsis as early as possible, even when a severe infection is not suspected. The answer may be leveraging an existing, routine test to include an indication of sepsis. By enhancing the most common laboratory test ordered by emergency physicians, a complete blood count (CBC) with an early sepsis indication may allow antibiotics to be administered sooner while not adding new burdens to physicians, nurses, laboratories, or the patient.
Current tests
The challenges surrounding sepsis diagnosis have led to sepsis commonly being under-or over-diagnosed.10 Screening of suspected sepsis patients includes basic vitals—heart and respiratory rates, oxygen saturation levels, blood pressure, and temperature. Because septic patients often show signs of change in mental status7 and speech patterns,11 these are also evaluated. The Systemic Inflammatory Response Syndrome (SIRS) criteria were introduced in 1992 as a tool to help diagnose sepsis. Under SIRS, diagnosis of sepsis requires the presence of two or more of four basic criteria (tachycardia, tachypnea, hyperthermia, or hypothermia (>38°C or <36°C) and changes in white blood count levels).12 In 1994, the Sequential Organ Failure Assessment (SOFA) score, based on platelet counts, creatinine levels, respiration, cognition, and liver and renal changes, was introduced as a way to describe the degree of organ failure of critically ill patients.13
In 2016, an international task force introduced the newest definition for sepsis: the Sepsis-3 (Sep-3) criteria. Under the Sep-3 criteria, sepsis is an infection with two or more of the previously defined SOFA points, whereas septic shock is sepsis with vasopressor-dependent hypotension and a lactate level greater than two.14 The same task force also introduced the quick SOFA (qSOFA) criteria to identify patients at high risk for a poor outcome. qSOFA is a simple test with only three components: respiratory rate >22 breaths/min, altered mental state, and systolic blood pressure <100 mmHg.15 Each component receives one point, and a score >2 has been found to be predictive of all-cause mortality outside of the ICU.15 While widely used in clinical practice for prognosis of septic patients, unfortunately, both SIRS and qSOFA have been shown to have a poor diagnostic value for sepsis.16, 17, 18In addition to the patient’s physical traits, physicians rely heavily on the laboratory for testing to try to pinpoint sepsis. Blood tests, including the complete blood count with differential (CBC-Diff), sputum testing, and urinalysis are commonly done in the ED to look for sepsis. Procalcitonin (PCT) is produced in response to bacterial infection, and high PCT levels can be indicative of a serious infection.19 Similarly, levels of C-reactive protein (CRP), a substance produced in the liver, increase in response to systemic inflammation or infection.20 While CRP has been shown to be very sensitive, it is not at all specific; increased levels can indicate, among other things, infection, cancer, or heart disease.
The variable nature of sepsis diagnosis,21 especially early in disease, and the poor predictive value of existing criteria17 and aforementioned biomarkers highlight the importance of the availability of a unique, early sepsis biomarker to aid clinicians in escalating or de-escalating treatment for patients in the ED. While hundreds of studies have been published on promising sepsis biomarkers, implementing them in clinical practice in already overworked and understaffed emergency departments (EDs) and clinical laboratories has been challenging.
A valuable sepsis biomarker needs to flag when risk of sepsis is present, not when it is not. It should be easy to collect, inexpensive, and done on patients regardless of previous sepsis suspicion. Recently, there has been renewed interest in using components of the CBC-Diff to find indicators of sepsis. The CBC-Diff is the most ordered test in the ED and comprises 85% of all blood tests ordered in the ED.22 It is easy to perform, has a rapid turnaround time, and provides a wealth of health information aiding in a differential diagnosis. Three CBC tests: red cell distribution width (RDW), neutrophil-to-lymphocyte ratio (NLR), and monocyte distribution width (MDW) have been shown to have potential as biomarkers for sepsis.
RDW is an erythrocyte index and reflects heterogeneity in the size of circulating RBCs. When used in patients with suspected sepsis, RDW has been shown to have modest value for predicting a positive blood culture, but limited value for diagnosing sepsis.23 Likewise, NLR has prognostic value similar to that of C-reactive protein.24 In times of physiological stress, neutrophil count increases and lymphocyte count decreases. The decrease in lymphocyte levels in early sepsis has been correlated with poorer outcomes in septic patients,25,26 but neutrophil levels can be influenced by chronic health conditions27 and lymphocyte levels can be decreased by certain syndromes and noninfectious health conditions28 confounding the ratio.
The value of MDW
Monocytes are the body’s first line of defense against pathogens. Part of both the innate and acquired immune responses, monocytes are activated in response to pro-inflammatory signals from infectious organisms and pathogen-associated molecular patterns (PAMPs) such as lipopolysaccharide. Monocytes express receptors for PAMPs and respond with transcriptional changes and downstream signaling that ultimately recruit leukocytes to the affected area.29 As with the size variability of RBCs, the monocytes’ response to infection causes an acute change in the size distribution.30 The monocyte distribution width (MDW) parameter, a regulatory-cleared, in vitro diagnostic measurement, reflects a change in the volume of circulating monocytes (See Figure 2).
The initial feasibility study on the diagnostic value of the MDW biomarker in adult patients in the ED showed that it alone or in combination with the WBC count could be used to help establish the severity of infection and risk of sepsis in the ED.33 When used in conjunction with other clinical parameters, the MDW biomarker has been shown to improve the early detection of sepsis during the initial ED encounter.31 And while the exact molecular mechanism for changes in MDW is unknown, it is possible that variations in MDW biomarker correspond with the shift from a localized infection to a systemic, septic inflammatory response.33
One of the biggest benefits of the MDW biomarker is that it is part of the CBC-Diff. There are no additional testing requirements, thus no impact to workflow. The clinical laboratory is central to patient care, and workflow must be executed in a way that ensures the most accurate results reach the clinicians in the most efficient way possible. Results from MDW biomarker studies demonstrated that test results are comparable to those of lactate and C-reactive protein (CRP).31,32 But as part of routine CBC testing, the MDW parameter was run in all patients whereas CRP and lactate were only run for patients with suspected sepsis. Thus, more patients received more information with lower impact to the lab.
The overwhelming dysfunctional immune response to infection leading to sepsis begins long before clinical symptoms are apparent. Having a unique biomarker that is evaluated as part of a standard test is a valuable tool to help detect patients at risk of developing sepsis.
Implementing MDW at Christus Trinity Mother Frances Health System
Earlier this year, the team at Christus Trinity Mother Frances Health System in Tyler, TX implemented the MDW parameter in seven locations — hospitals and standalone emergency departments — to aid in early sepsis detection knowing that strategically adding MDW to the EDs could be a game changer for sepsis outcomes. When patients enter the ED, they are a blank slate. Other than vital signs and the often-limited information provided by patients, very little is known. MDW allows every adult patient to be screened — not just those with specific sepsis symptoms — without interrupting the workflow in the lab or ED. The physician doesn’t have to be looking for sepsis, but this allows for earlier identification. In the first week of use, the Christus team used the MDW biomarker to detect early sepsis in a patient; she was treated within two hours, and as a result, had a hospital stay of less than two days. When sepsis is diagnosed earlier, treatments can be started sooner, which leads to better patient outcomes. And achieving better patient outcomes is really what it’s all about.
Closing thoughts
Monocytes react early in infection, so MDW provides an opportunity for clinicians to narrow differential diagnosis at the beginning of the patient encounter. Because the MDW parameter is measured as part of the CBC-Diff, it has value for identifying the possibility of sepsis in individuals for whom sepsis is not immediately suspected without adding to the already too-heavy workflow of the lab. The MDW parameter is not intended as a replacement for the qSOFA and SIRS criteria clinical analyses, but rather as an addition to these screening parameters to improve early sepsis detection.33 Combined with innovative approaches to treatment, the MDW biomarker helps to identify severity of infection and sepsis, which may help to reduce the mortality rate associated with sepsis. Early sepsis detection leading to early source control can literally be the difference between life and death.
“Because the MDW is available in the CBC with differential, it’s available much earlier than the other biomarkers. So MDW, in combination with other patient information, will help improve our ability to identify septic patients and improve outcomes.” ~Dr. Freimer
References
- Rudd KE, Johnson SC, Agesa KM, et al. Global, regional, and national sepsis incidence and mortality, 1990–2017: analysis for the Global Burden of Disease Study. The Lancet. 2020;395(10219):200-211. doi:10.1016/s0140-6736(19)32989-7.
- Rudd KE, Kissoon N, Limmathurotsakul D, et al. The global burden of sepsis: barriers and potential solutions. Critical Care. 2018;22(1). doi:10.1186/s13054-018-2157-z.
- Kumar G, Kumar N, Taneja A, et al. Nationwide Trends of Severe Sepsis in the 21st Century (2000–2007). Chest. 2011;140(5):1223-1231. doi:10.1378/chest.11-0352.
- National Center for Health Statistics. NCHS Data Brief, Number 62, June 2011.; 2011. Accessed September 26, 2022. http://www.cdc.gov/nchs/data/databriefs/db62.pdf
- Kumar A, Roberts D, Wood KE, et al. Duration of hypotension before initiation of effective antimicrobial therapy is the critical determinant of survival in human septic shock. Crit Care Med. 2006;34(6):1589-1596. doi:10.1097/01.CCM.0000217961.75225.E9.
- Peltan ID, Brown SM, Bledsoe JR, et al. ED door-to-antibiotic time and long-term mortality in sepsis. Chest. 2019;155(5):938-946. doi:10.1016/j.chest.2019.02.008.
- Centers for Disease Control and Prevention. What is sepsis? Centers for Disease Control and Prevention. Published January 27, 2021. https://www.cdc.gov/sepsis/what-is-sepsis.html Accessed September 22, 2022.
- Filbin MR, Lynch J, Gillingham TD, et al. Presenting symptoms independently predict mortality in septic shock: Importance of a previously unmeasured confounder. Crit Care Med. 2018;46(10):1592-1599. doi:10.1097/CCM.0000000000003260.
- Dunne D, McDonald R, Ratnayake R, et al. Blood cultures in acute surgical admissions. Annals of The Royal College of Surgeons of England. 2015;97(1):27-31. doi:10.1308/003588414X14055925059110.
- Confield LR, Black GP, Wilson BC, Lowe DJ, Theakstone AG, Baker MJ. Vibrational spectroscopic analysis of blood for diagnosis of infections and sepsis: a review of requirements for a rapid diagnostic test. Analytical Methods. 2021;13(2):157-168. doi:10.1039/d0ay01991g.
- Wattanapaiboon K, Banditlerdruk S, Vattanavanit V. Presenting symptoms in sepsis: Is the mnemonic “SEPSIS” useful?. Infect Drug Resist. 2020;13:2199-2204. Published 2020 Jul 8. doi:10.2147/IDR.S263964.
- Comstedt P, Storgaard M, Lassen AT. The Systemic inflammatory response syndrome (SIRS) in acutely hospitalised medical patients: a cohort study. Scand J Trauma Resusc Emerg Med. 2009;17(1):67. doi:10.1186/1757-7241-17-67.
- Lambden S, Laterre PF, Levy MM, Francois B. The SOFA score—development, utility and challenges of accurate assessment in clinical trials. Critical Care. 2019;23(1). doi:10.1186/s13054-019-2663-7.
- Marik PE, Taeb AM. SIRS, qSOFA and new sepsis definition. J Thorac Dis. 2017;9(4):943-945. doi:10.21037/jtd.2017.03.125.
- Seymour CW, Liu VX, Iwashyna TJ, et al. Assessment of clinical criteria for sepsis. JAMA. 2016;315(8):762. doi:10.1001/jama.2016.0288.
- Askim Å, Moser F, Gustad LT, et al. Poor performance of quick-SOFA (qSOFA) score in predicting severe sepsis and mortality - a prospective study of patients admitted with infection to the emergency department. Scand J Trauma Resusc Emerg Med. 2017;25(1):56. doi:10.1186/s13049-017-0399-4.
- Tusgul S, Carron PN, Yersin B, Calandra T, Dami F. Low sensitivity of qSOFA, SIRS criteria and sepsis definition to identify infected patients at risk of complication in the prehospital setting and at the emergency department triage. Scand J Trauma Resusc Emerg Med. 2017;25(1):108. Published 2017 Nov 3. doi:10.1186/s13049-017-0449-y.
- Jouffroy R, Saade A, Carpentier A, et al. Triage of septic patients using qSOFA criteria at the SAMU regulation: A retrospective analysis. Prehosp Emerg Care. 2018;22(1):84-90. doi:10.1080/10903127.2017.1347733.
- Pierrakos C, Velissaris D, Bisdorff M, Marshall JC, Vincent JL. Biomarkers of sepsis: time for a reappraisal. Crit Care. 2020;24(1):287. Published 2020 Jun 5. doi:10.1186/s13054-020-02993-5.
- Póvoa P, Almeida E, Moreira P, et al. C-reactive protein as an indicator of sepsis. Intensive Care Med. 1998;24(10):1052-1056. doi:10.1007/s001340050715.
- Rhee C, Kadri SS, Danner RL, et al. Diagnosing sepsis is subjective and highly variable: a survey of intensivists using case vignettes. Critical Care. 2016;20(1). doi:10.1186/s13054-016-1266-9.
- Rui P, Kang K. National Hospital Ambulatory Medical Care Survey: 2017 emergency department summary tables. National Center for Health Statistics. Available from: https://www.cdc.gov/nchs/data/nhamcs/web_tables/2017_ed_web_tables-508.pdf. Accessed November 12, 2022.
- Hu ZD, Lippi G, Montagnana M. Diagnostic and prognostic value of red blood cell distribution width in sepsis: A narrative review. Clin Biochem. 2020;77:1-6. doi:10.1016/j.clinbiochem.2020.01.001.
- Drăgoescu, A. et al. (2021). Neutrophil to lymphocyte ratio (NLR)-A useful tool for the prognosis of sepsis in the ICU. Biomedicines. 10(1), 75. https://doi.org/10.3390/biomedicines10010075.
- Venet F., Davin F., Guignant C., Larue A., Cazalis M.A., Darbon R., Allombert C., Mougin B., Malcus C., Poitevin-Later F., et al. Early assessment of leukocyte alterations at diagnosis of septic shock. Shock. 2010;34:358–363. doi: 10.1097/SHK.0b013e3181dc0977.
- Wesche D. et al. (2005). Leukocyte apoptosis and its significance in sepsis and shock. J. Leukoc. Biol. 78:325–337. doi: 10.1189/jlb.0105017.
- Belok S.H., Bosch N.A., Klings E.S., Walkey A.J. Evaluation of leukopenia during sepsis as a marker of sepsis-defining organ dysfunction. PLoS ONE. 2021;16:e0252206. doi:10.1371/journal.pone.0252206.
- Lymphopenia - Causes and Risk Factors | NHLBI, NIH. www.nhlbi.nih.gov. Accessed September 28, 2022. https://www.nhlbi.nih.gov/health/lymphopenia/causes.
- Mogensen TH. Pathogen recognition and inflammatory signaling in innate immune defenses. Clinical Microbiology Reviews. 2009;22(2):240-273. doi:10.1128/cmr.00046-08.
- Crouser ED, Parrillo JE, Seymour CW, et al. Monocyte distribution width: A novel indicator of sepsis-2 and sepsis-3 in high-risk emergency department patients. Crit Care Med. 2019;47(8):1018-1025. doi:10.1097/CCM.0000000000003799.
- Crouser ED, Parrillo JE, Martin GS, et al. Monocyte distribution width enhances early sepsis detection in the emergency department beyond SIRS and qSOFA. J Intensive Care. 2020;8(1). doi:10.1186/s40560-020-00446-3.
- Malinovska A, Hinson JS, Badaki-Makun O, et al. Monocyte distribution width as part of a broad pragmatic sepsis screen in the emergency department. J Am Coll Emerg Physicians Open. 2022;3(2):e12679. doi:10.1002/emp2.12679.
- Crouser ED, Parrillo JE, Seymour C, et al. Improved early detection of sepsis in the ED with a novel monocyte distribution width biomarker. Chest. 2017;152(3):518-526. doi:10.1016/j.chest.2017.05.039.
About the Author

Patusa Mayfield, MBA-HCM, MT(ASCP)
is Director of Laboratory Services at Christus Trinity Mother Frances Health System, Tyler, Texas.