Operational efficiency, financial visibility, and workforce pressures shape laboratory best practices in 2026
Medical laboratories continue to operate under mounting pressure to balance operational efficiency, financial stability and quality performance. Results from the 2026 MLO State of the Industry (SOI) survey on lab management best practices highlight how labs are prioritizing process improvement and cost control while navigating persistent challenges with reimbursement visibility and system integration.
One of the most notable findings is the emphasis on operational efficiency. Nearly two-thirds of respondents (63%) said technology investments are primarily aimed at improving efficiency or reducing costs, while 56% reported prioritizing replacement of aging equipment. Automation remains central to operational performance, with 40% of laboratories adopting analyzers that enable walkaway testing to reduce staff demands.
At the same time, financial visibility remains a concern. Only 32% of respondents said they can track whether reimbursement fully covers their testing costs, down from 33% in 2025 and 45% in 2024. Data integration appears to be a major stumbling block, with 33% of laboratory professionals citing lack of interoperability between laboratory information systems (LIS) and revenue cycle platforms as barriers to tracking reimbursement levels.
Operational performance metrics also remain central to laboratory management strategies, with 63% of respondents reporting they regularly review turnaround time to identify improvement opportunities.
Also new to this year’s survey, MLO asked laboratory professionals a series of questions on proficiency testing, revealing a mix of traditional manual processes and growing use of proficiency testing data for training, quality improvement, and identifying instrument or method bias.
The respondents
This year, MLO gathered responses from 362 medical laboratory professionals, with 41% in director, manager, administrator, or supervisor positions and most employed by hospital/health system laboratories (66%). Respondents again represented a broad range of laboratory sizes and testing volumes.
Workforce
Recognizing the ongoing staffing challenges facing U.S. laboratories, MLO asked medical laboratory professionals about the strategies they are using to retain and recruit staff. Continuing education topped the list at 46% (same as 2025). This was followed closely by a new answer option for this year’s survey—team building/celebration activities (43%).
Leveraging shift changes to offer scheduling flexibility (e.g., 12-hour shifts, weekends) increased in popularity at 39%, up from 33% in 2025.
While financial incentives remained high on the list with 37% of respondents citing it as a strategy, it dropped from 48% in 2025. The percentage of laboratory professionals citing partnerships with local colleges and tech schools to offer internships also dropped at 37% in 2026, down from 46% last year.
Daily huddles with peer recognition remained a popular recruitment and retainment strategy, at 35% of respondents (36% in 2025), along with clinical ladders at 32% (33% in 2025), utilizing outside laboratory recruitment services/agencies at 24% (26% in 2025) and succession-planning processes at 20% (22% in 2025).
Offering employee perks (free parking, onsite gym, onsite day care, reimburse public transportation costs) remained the least cited strategy at 16% (17% in 2025).
Sheridan M. Voshake, MS, MLS(ASCP)cm, laboratory manager for Memorial Hospital and Adjunct Professor at Carl Sandburg College, both located in Carthage, Ill., offered her insights on laboratory workforce challenges today – and best practices for addressing them:
“Recruitment and retention remain challenging, particularly as medical laboratory training programs continue to close, reducing the pipeline of qualified professionals entering the field. Today’s candidates are prioritizing workplace culture, flexibility, and meaningful opportunities for growth. We are intentionally focused on strengthening employee engagement and supporting career advancement with pathways from phlebotomists to medical laboratory technicians and medical laboratory scientists. This educational commitment has been instrumental in driving stronger retention outcomes within our laboratory department and organization.”
Reimbursement
When asked whether their laboratories had taken steps to ensure reimbursement covers testing costs, the top response was standardization strategies, with 53% of respondents citing standardized instrumentation workflows and checklists (53% in 2025) and 53% citing the creation of standard lab processes and staff education materials (52% in 2025).
A focus on efficiency and accuracy was evidenced by 40% of respondents naming the adoption of analyzers that provide walkaway testing to reduce staffing and FTE hours as a strategy to balance costs and reimbursement (37% in 2025) and 36% naming the use of IT solutions to reduce human error (46% in 2025).
New for this year, MLO queried laboratory professionals on whether they evaluate reimbursement for all participating insurance providers to ensure rates will cover costs before implementing a test. Over one-third of respondents (33%) acknowledged leveraging this approach.
Other steps laboratories reported taking to ensure reimbursement covers costs include:
- 24% incorporating IT solutions to help keep current with regulations
- 23% adopting processes to review savings opportunities, such as evaluating analyzers, on a regular schedule
- 20% implementing ongoing waste and efficiency studies to find potential savings in overhead
- 19% bringing health screening tests in house
- 13% implementing ongoing efforts to reduce coding frustrations and modifications
Only 32% of laboratory professionals surveyed indicated their laboratories could ensure reimbursement covers their costs. The biggest stumbling block cited was lack of interoperability between laboratory information system (LIS) and revenue cycle management software at 33%. This was followed closely by lack of staff time to perform this task at 30%.
The focus on inadequate technology and staffing was also reflected in the high ranking of other stumbling blocks: 29% do not have software to automate tracking/analysis of costs and 25% don’t have enough IT staff time/resources.
Lastly, 6% of respondents cited not enough barcoded testing supplies as a challenge to tracking whether reimbursement covers their costs.
LigoLab RCM Product Manager Aram Avakyan commented on how laboratories are leveraging modern LIS platforms to improve operational visibility across testing, billing, and reporting workflows:
“Modern laboratory information systems enhance operational visibility by using configurable rules and workflow tags to track activity across the laboratory process. As a case or specimen moves through the lab, the LIS can apply tags that indicate status, progress, exceptions, or required follow-up actions, allowing teams to quickly see where work is occurring and where delays may exist. Because each tag captures structured workflow data, laboratories can also use them for reporting and operational analysis to identify bottlenecks and monitor key performance metrics.”
“Modern LIS platforms provide laboratories the data visibility needed to connect operational activity with financial performance,” Avakyan added. “One of the most important capabilities is flexible reporting and analytics, including preconfigured reports that track metrics across testing activity, billing status, and reporting performance. Systems that allow labs to create dynamic reports tied to workflow data also make it easier to identify revenue gaps, monitor payer behavior, and understand how operational decisions affect reimbursement outcomes.”
Test quality and efficiency
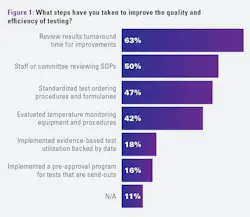
Nearly two-thirds of laboratory professionals (63%) reported reviewing turnaround time to identify opportunities to improve test quality and efficiency. This was a new question answer option provided this year.
Other popular strategies to improve test quality and efficiency are staff or committee reviews of standard operating procedures (SOP) (50%), leveraging standardized test ordering procedures and formularies (47%), and evaluating temperature monitoring equipment and procedures (42%).
Fewer cited the implementation of evidence-based test utilization backed by data (18%) or pre-approval programs for tests that are send-outs (16%).
Maine Molecular Quality Controls (MMQCI) Manager of Discovery Dr. Stehen Pelsue, Ph.D. commented on the quality control challenges most common today in molecular or specialized testing, and how can laboratories strengthen validation and ongoing monitoring:
“QC challenges for molecular assays can be driven by rapid technology development and complex assay targets that evolve faster than available guidance and availability of proper controls. External quality assessment options remain sparse for many NGS LDTs, particularly for rare or complex variants. End to end validation should evaluate all specimen types and pre analytic steps, laboratory process, and the bioinformatics workflow, with documented sensitivity/specificity for each relevant variant type, as not all variants may be available.”
"Labs can use QC performance data strategically to improve reliability, not just as a compliance requirement,” he added. “For example, monitoring longitudinal QC data for gradual performance drifts instead of reacting only to out‑of‑range calls. Evaluating trends over time can identify subtle shifts linked to lot changes, instrument issues, or environmental conditions before they affect patient results.”
Proficiency testing
This year’s MLO SOI on laboratory best practices explored a new topic: proficiency testing. Respondents were asked how their laboratories ensure all required analytes and methods are appropriately enrolled in proficiency testing. The majority (77%) responded with manual review of test menu.
Responses on other ways to ensure all required analytes and methods are appropriately enrolled in proficiency testing:
- 17% PT provider recommendations
- 13% external consultant/accreditation support
- 9% middleware or analytics software
- 8% LIS-based test mapping
- 8% unsure/unclear
- 7% other
Laboratory professionals were asked how their laboratories track and analyze proficiency testing performance over time. The most common response was reviewing manually after failures (62%), followed by trending across multiple events (51%).
Over one-third of survey respondents selected event-by-event review only (38%) or integrated into QA dashboards (35%), while 7% reported their proficiency testing performance was not routinely analyzed.
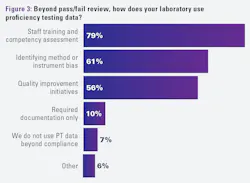
The final question on this topic asked how their laboratories use proficiency testing data beyond pass/fail review. Over three-quarters of respondents (79%) reported using this data for staff training and competency assessment, followed by identifying method or instrument bias (61%) and quality improvement initiatives (56%). Far fewer selected required documentation only (10%), while 7% reported not using PT data beyond compliance.
WSLH Proficiency Testing Lab Associate Director Tracy Servey, MT (ASCP) commented on practices that can help labs move beyond simply meeting proficiency testing requirements to using results as a tool for continuous improvement:
“We see lab leaders focus on the final evaluation scores and ignore the other details that accompany the report. For example, the standard deviation Index (SDI) should not be ignored when reviewing quantitative results on the evaluation report. That is, although your evaluation report may list a passing score, a positive or negative bias reflected by your SDIs should be investigated to avoid potential systemic errors.”
When what common gaps or misconceptions exist in how laboratories select, manage or interpret proficiency testing programs—and how can organizations strengthen their approach—Servey stated:
“There is a balance between PT program scope and laboratory need. With different types of testing devices entering the market, test menus between different laboratories have become far more diverse but the PT programs may have limitations in offerings out of necessity. Time should be taken to assure the lab’s test menu is aligned with the PT programs being used to assure the test instruments are being fully challenged.”
Technology
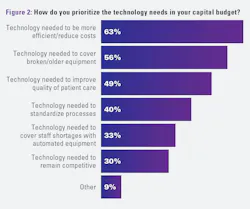
To better understand which factors influence new technology investments, MLO asked laboratory professionals how they prioritize the needs for their capital budgets. The most reported factor was technology needed to be more efficient/reduce costs (63%).
The next top response was technology needed to cover broken/older equipment (56%), followed by technology needed to improve quality of patient care (49%).
The need for standardization and efficiency was again reflected in the next two factors on the list: technology needed to standardize processes (40%) and technology needed to cover staff shortages with automated equipment (33%). A further one-third of respondents cited technology needed to remain competitive (30%).
When examining best practices for adopting new tools for laboratory automation, the responses were similar to last year’s survey, with nearly half of respondents reporting they analyzed workflow processes for proper space planning (46%, up from 44% in 2025), 39% involved the IT department early in the process (40% in 2025), 30% ensured system integration for seamless process and data flows (36% in 2025) and 18% designated a project manager to coordinate short- and long-term planning and implementation with the vendor (21% in 2025). More than one-third (36%) selected N/A.
With regards to training staff on new software, the most reported best practice by laboratory professionals was the creation of standard workflows for all lab employees (61%). Nearly half reported creating a train-the-trainer model (48%).
More than one-third reported using vendor-hosted in-house or online training (34%), an annual online education refresher (32%) or LIS training of a lab staff member to develop an in-house expert (32%). Other best practices for training staff on new software included developing mandatory training for new lab employees led by the IT department (20%) and lunch-and-learn training sessions (16%).
Contracting and supply chain
Laboratory professionals were asked what best practices they had implemented to streamline their vendor contracting process. The highest percentage of respondents (65%) reported working with supply chain management on supplies that are on group purchasing organization (GPO) contracts that offer additional savings.
The second most commonly reported best practice for vendor contracting (40%) was the development of good relationships with supplier support personnel that provide access to training and product optimization suggestions. This was followed by adoption of ongoing reviews of reference lab costs and contracts (35%) and the signing of longer contracts (20%).
The best practice that topped the list for improving inventory control and consumable supply costs was the evaluation of inventory levels for basic supplies, such as assays and controls/reagents (66%). More than one-third of laboratory professionals (38%) reported developing supply utilization tracking and record keeping.
Responses on other ways to improve inventory control and consumable supply costs:
- 19% secured access to electronic inventory tracking from the supply chain/materials management department
- 18% implemented lease agreements that do not include volume commitments
- 17% worked with other members of the organization, such as the CMO (Chief Medical Officer) and physicians, to standardize test ordering throughout the organization
- 16% developed ongoing review comparing supply reports to the number of invoiced tests
- 13% implemented vendor-managed ordering
- 11% this is handled by a different organization/location
Looking at best practices to address supply chain issues, the most reported practices were implementing standing orders (instead of just in time) for crucial supplies (54%) and establishing relationships with multiple vendors (50%).
More than one-third of laboratory professionals reported they are not currently experiencing supply chain issues 34%.
Reported use of several best practices have declined: utilizing multiple testing platforms (20% in 2026, down from 27% in 2025, 30% in 2024), laboratory-developed tests (LDT) (6% in 2026, down from 13% in 2025), working with state public health officials to gain access to needed testing supplies (5% in 2026, down from 12% in 2025 and 16% in 2024).
The best practice of switching to reusable types of personal protective equipment, such as moving from disposable to reusable lab coats, increased only slightly year-over-year (17% in 2026, 16% in 2025, 20% in 2024).
Conclusion
Taken together, the survey findings suggest laboratories are increasingly focused on operational efficiency, financial visibility, and workforce sustainability. As laboratories continue adopting automation and data-driven management practices, addressing interoperability, reimbursement tracking, and workforce development will remain critical priorities in the years ahead.
About the Author

Kara Nadeau
has 20+ years of experience as a healthcare/medical/technology writer, having served medical device and pharmaceutical manufacturers, healthcare facilities, software and service providers, non-profit organizations and industry associations.