Moving clinical departments to new facilities can be practical in today’s healthcare service for various reasons, but the relocation of an essential laboratory department had not occurred at Rush University Medical Center in over 35 years. The aim of moving the Blood Center from its previous location—to a location that is more proximal to critical care units—was to provide improved efficiency and allow reduced transport time of blood components. The development plan for the relocation began in 2016, which took two years to design and required regular site visits early on in the project. Relocating a laboratory department to a new location involved modifications of operational procedures and construction of the physical design. A strategic management plan, an efficient schedule, and overall teamwork was required to sustain continuity of quality patient care and ensure the transition was successful. This article will describe the various stages of planning that were needed in order to successfully relocate the services of the Blood Center, and to describe some of the experiences and lessons the clinical department learned during the process.
Introduction
Since 1980, the Blood Center had operated out of a 15-story building that was built in 1960. Known as the Jelke Building, the previous site of the Blood Center also housed the former Blood Donor Center and Apheresis Unit. The Jelke Building accommodated the Neonatal Unit and Labor and Delivery Suites until the opening of the new patient building, referred to as “the Tower.” The new Tower, located on the opposite end of the medical facility, was completed in December of 2011, and patients first moved in on January 2012. The volume of patients seen at Jelke has since decreased and many services have been discontinued.
Planning to move the Blood Center from Jelke to the Tower began in 2012. The space that was identified for the new lab was previously being used as a training area that contained mock patient rooms for the future development of an ambulatory care clinic. It was not until 2016 that the project plan and budget had been approved by hospital administration, and the initial blueprint was created.
One of the top priorities of the move was to continue the mission of the hospital in providing enhanced services and high-quality care. By moving the Blood Center out of Jelke and into a centrally located space within the Tower, positioned services much closer to critical care areas, thereby reducing waste and decreasing the transport time of blood products from the laboratory and to the patients.
Planning the move
The major stakeholders who guided the development of the new lab included Capital Projects executives, administrative directors, the Blood Center medical director and manager, pathology administrators, shift supervisors, the quality assurance coordinator, and Laboratory Information System (LIS) team. Weekly internal team meetings were held to address the construction status, equipment purchasing, and the area floorplan. The project leaders, alongside the architecture firm, worked closely on the arrangement of key parts within the lab. Planning began with a rearrangement of the lab’s work benches, in order to determine how each structure could maximize operational efficiency and conform to Lean Six Sigma principles. Meetings were then held with the Blood Center staff on an as-needed basis for several weeks regarding their input with the overall lab design.
An open house was arranged to increase staff awareness of the Blood Center’s transition. Prior to the open house, the lab had been subdivided into specific workbenches, including:
1. Expediting: responsible for receiving specimens and dispensing products to the floor.
2. Type and Screen: responsible for running all patient samples on an automated gel-card analyzer.
3. Crossmatch Stations 1, 2, and 3: each responsible for completing transfusion orders for all blood products.
4. Consultation: responsible for performing antibody identification.
5. Miscellaneous space for component preparation for pediatric aliquots, washed, and/or irradiated products.
Official tours during the open house were given by the Blood Center manager and visual cues were arranged to reflect where equipment and supplies were to be placed on each bench.
In the month preceding the official move date, staff assignments took place for validation/requalification purposes to ensure all equipment and supplies were successfully placed by the go-live date. These key responsibilities were all included as part of the Tower Move Manual and served as a guide provided by the Blood Center manager. A communication plan was provided for any unexpected difficulties that occurred and incorporated the phone numbers of the incident commanders, logistics/operations contacts, as well as the directory of other important phone numbers including our main blood supplier, LIS on-call, gel card analyzer service support, hospital transport services, and medical center engineering, among others. Standard operating procedures were drafted to reflect new workflows within the Tower.
The Tower Move Manual also contained a timetable for the move-in weekend, outlined bench responsibilities, scheduled inventory delivery dates, workflow procedures, and a phone directory for each bench. Responsibilities were divided between first and second shift Blood Center staff, and had to be completed by the timeline indicated in the Tower Manual.
Before the equipment was physically moved out of Jelke, all important pieces of equipment that were needed on the bench, such as centrifuges and cell washers, were first drafted on a cardboard template and appropriately labeled to reflect where each piece would be situated on the counter space.
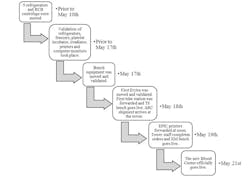
The original Blood Center move date (May 1) was delayed due to an inspection by the Illinois Department of Public Health. Nonetheless, preparation continued as the new date was in the process of being finalized. Each piece of critical equipment, beginning with the heaviest to transport, was labeled with a moving date to ensure all staff was fully aware of its removal. This provided time to allow any requalification or revalidation to take place in its new location. All of the newly purchased equipment, including four freezers, a platelet incubator, and an irradiator, was scheduled to be delivered to the Tower location. The initial validation process not only involved critical equipment, but also included the validation of new printers, computer monitors, and barcode scanners by the LIS specialist prior to the official move-in date. Coordination of the move did not solely involve the project leaders, but it also relied on the gel card analyzer technical specialists who played a crucial part in relocating and reprogramming the instruments.
Part of the communication plan pertaining to the hospital wide notification of the Blood Center’s relocation occurred via email on May 16. Our external partners, including the outpatient centers and affiliated hospitals, were also notified via email. A few weeks prior, the capital projects department had modified the hospital directory and facility signs to include the Blood Center. An announcement was also published on the medical center’s intranet, a valuable tool to inform all hospital staff of its new transition. A memo was sent out to nursing leadership regarding the new tube station number and location, with the goal of disseminating this information to the inpatient and outpatient nursing staff.
The first step of the move took place on Friday, May 18, where the first gel card analyzer was relocated to the Tower, adjacent to the Type and Screen bench. Two of the analyzer technical specialists had arrived to properly shut it down and the movers hired by Capital Projects, delivered the instrument to the new Blood Center shortly after. The second gel card analyzer remained in the Jelke building and continued running samples during this time. After several hours, the analyzer was set up and functioned to begin the revalidation process. Despite some miscommunication with medical center engineering, which had begun re-routing the tube station earlier than anticipated, the instrument was finally equipped to run samples after successful completion of the validation. In the afternoon, both tube stations were re-routed and the Type and Screen bench officially went live. Once Type and Screen were discontinued in Jelke, the only active remaining benches were Crossmatch 1 and 2, which continued to fulfill all hold/transfusion orders. The Consultation bench completed the antibody identification workups. The Expediting bench was also relocated to the Tower to process incoming specimens after the re-routing of the tube stations. A command center was set up in the Blood Center conference room and team members were accessible to respond to any issues that might arise to avoid overwhelming staff and provided a telephone directory in case they were unavailable. Continuous communication between both operating labs avoided confusion and nervous tension, despite having two of the benches in completely separate areas and half of the equipment missing.
While most of the refrigerators were already transferred to the new lab, around half of the inventory levels of red cells and platelets were kept in the remaining fridges and platelet incubator. The blood supplier began delivering inventory orders to the new lab on the first day of the move. In order to transfer the current inventory of red cells, plasma products, and frozen red cells, blood supplier delivery boxes and dry ice were used for proper storage.
On the second day of the move, the printers were forwarded at noon to have all hold and transfuse orders print out from those located in the Tower. To minimize the risk of discrepancies between the orders that printed, the printers located in Jelke were never shut off, but instead printed at both locations. Once a confirmation was given that the printers were working effectively, the Crossmatch 1 bench went live on May 19 after some of the equipment including the heat blocks, centrifuges, and sterile connectors were ready for use.
While one technologist remained in Jelke, constant communication between Jelke and the Tower staff again ensured that all orders were being completed. Despite trying to accommodate sufficient coverage for the two operating Blood Centers, the schedule was rearranged to 12-hour work shifts between all Blood Center staff. By May 20 this “test environment” had been resolved from most deficiencies in order to prepare for the official go-live date. The new Blood Center officially opened on May 21.
Lessons learned
The construction of a new project is almost always interrupted by challenges and alterations to the original plan. While planning for the design of the new lab, one of the greatest disagreements came between Capital Projects, the architect, and Blood Center management. While the architect’s goal was primarily aesthetic, the Blood Center found its demands were not being met, which led to frustration and reorganization of the blueprint. A consultant experienced in the construction of laboratories would have been beneficial to help the architect and construction personnel acknowledge the requests of the lab and improve the structure of the design. Another major issue that occurred involved the phone installation for each bench. Ultimately, it took three days for these communication glitches to be resolved.
Conclusion
Relocating an active laboratory can be a challenging event for everyone involved. By following a structured plan and adhering to a well-organized timeline, the entire project successfully maintained two operating labs, safely moved all equipment, and minimized disruptions to patient care during the transition. Its new location within the hospital has optimized operations and has also improved accessibility for the critical care areas.
A special thank you to Miriam Miller, the Rush Blood Center Manager, for her assistance and insights of the relocation throughout the completion of this paper.