Common needs and interventions for COVID-19 and drug-resistant infections
For a printable version of the May CE Story and test go HERE or to take test online go HERE. For more information, visit the Continuing Education tab.
LEARNING OBJECTIVES
Upon completion of this article, the reader will be able to:
1. Describe how antimicrobial resistance occurs, what problems it causes, and examples of several drug-resistant organisms
2. Discuss why tracking of drug-resistant organisms is important and tracking tools available today
3. Discuss the role of infection prevention and control in preventing drug-resistant infections
4. Discuss the role of diagnostic testing in improving antibiotic stewardship
We began this year living in a world with a nearly untreatable virus. At no time in recent history has there been a series of events or circumstances that challenged organizations and individuals to engage in new actions and behaviors as exists today due to COVID-19. The COVID-19 pandemic has been an acute shock to the world, as the novel coronavirus has affected the health of millions and the lives and livelihood of billions.
This circumstance provides a glimpse of what the future is likely to become more broadly if we do not mobilize a sufficient global response to drug resistant infections caused by antimicrobial resistance (AMR), which occurs when drugs lose their effectiveness. It occurs when bacteria, viruses, fungi, and parasites change over time and no longer respond to medicines, making infections harder to treat and increasing the risk of disease spread, severe illness and death.1
While the urgent and immediate threat of the novel coronavirus is of utmost priority for governments and healthcare systems, we must ensure that the lessons we are learning today from COVID-19 shape continued actions to combat AMR now and in the years ahead. The global strategy to curb drug resistance, initiated in 2016 by the UN InterAgency Coordination Group on AMR,2 provides an effective framework for AMR action plans at the global and national level. The experience with the COVID-19 pandemic presents a unique opportunity to reexamine, adjust and reset our approaches through the lens of what actually happens when a nearly untreatable infection impacts the entire world.
Clinical and research laboratories have played a critical role in the COVID-19 pandemic. Laboratory testing results provide data to clinicians and allows patients to be put on the optimal treatment path. This data also informs surveillance, infection prevention practices, and antibiotic stewardship.
The impact of surveillance data
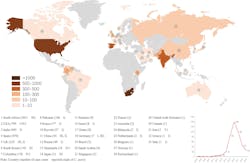
Tracking the spread of COVID-19 has been critical to the global public health response. This information allows hospitals to prepare for surges, governments to deploy testing strategies, and citizens to modify their behavior appropriately. But while information about COVID-19 is readily and publicly available, the world still struggles to monitor and track the spread of drug resistant infections.Candida auris is considered a “superbug fungus,” which has caused worldwide concern and has been classified as an Urgent Threat by the Centers for Disease Control and Prevention (CDC).4 C. auris is resistant to most first-line antifungal medications and spreads easily between patients. In 2020, Chen, et.al., completed a systemic review and meta-analysis of the global epidemiology and mortality of Candida auris. The analysis included studies from 2009 to 2019. Initially identified in Japan in 2009, there have been more than 4,733 cases of C. auris reported in more than 33 countries. Bloodstream infection was observed in 32% of the cases. The overall mortality of C. auris infection was 39%.5 This retrospective analysis shows the global spread of this pathogen and the critical need to have real-time surveillance to track these life-threatening pathogens.
Global reported cases of C. auris by country
As Jim O’Neill, author of the book, Superbugs: An Arms Race Against Bacteria, explains, “AMR is not a problem that can be solved by any one country, or even any one region. We live in a connected world where people, animals, and food travel, and microbes travel with them.”6
Using infection prevention and control to stop infections before they start
Preventing drug resistant infections reduces the use of antibiotics and improves patient outcomes. Infection control practices, from simple handwashing to global vaccination, and the use of effective infection prevention measures are key tools to combat COVID-19 and AMR.
Within healthcare systems, infection prevention measures are not specific to one pathogen and can have a broad impact. A recent study by Wee, et.al., evaluated the impact of a multimodal IPC strategy originally designed for the containment of COVID-19 on the rates of other hospital-acquired-infections (HAIs). With enhanced IPC measures introduced to contain COVID-19, they saw a decrease in hospital-wide MRSA acquisition rates together with central line-associated bloodstream infections rates. The rates of CRE, C. difficile infections, and device-associated HAIs remained stable. Respiratory infections were prioritized for interventions; however, good adherence to IPC impacted HAI rates as well.7
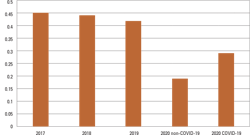
In a study by Bentivenga, et.al., researchers reported a significant reduction in the incidence of total MDR bacterial infections observed during the pandemic compared to in pre-pandemic years (p < 0.05). The study concluded that maintaining a high level of preventive measures could help tackle an important health problem, such as the spread of HAIs and multidrug resistant bacteria (MDRB).8
Incidence of MDR bacteria pre and post COVID-19Diagnostics are critical to slowing the spread of AMR. Better diagnostics can guide faster, more appropriate treatment to ensure patients receive the right drug for their infection, at the right time, for the right duration. Diagnostics can help us to preserve antibiotics and ensure they remain effective when they are needed most.
Dedicated funding and strong market need can and have driven rapid innovation. As we’ve seen, COVID-19 diagnostic tests were developed within weeks of the outbreak along with public health capabilities to address the pandemic like digital data collection, local manufacturing, and global distribution. There have been platforms built to collate data, resources marshaled, and creativity unleashed. We should capitalize on this innovation for AMR across the board, but we should certainly continue to move diagnostics for antibiotic-resistant pathogens forward.
The role of cross sector collaboration
Governments, researchers, and industry partners around the world have mobilized to develop diagnostics, therapeutics, and vaccines for COVID-19. The development of new recommendations and practices continue to be deployed to slow the spread of COVID-19. The burden of drug-resistant infections will likely surpass COVID-19. Drugs like antibiotics are a vital tool in modern medicine to prevent and treat infections. As drug-resistant infections are becoming more common, modern medicine as we know it is at risk, and much like COVID-19, these infections have the potential to overwhelm our healthcare structure.
In an article by Williams, et.al., the authors state that antimicrobial resistance is a threat to global health and food security. The emergence of COVID-19 in 2020 has focused attention on yet another global health challenge. The difference is that the pandemic is seen as a proximate public health crisis requiring immediate action. Consequently, relatively less attention has been directed towards antimicrobial resistance and climate change, which also pose urgent threats to lives and livelihoods, and in the end, may have worse global consequences.14
There are several tools available to learn more about how a cross sector approach will drive an improved focus, leading to a positive impact on the risk of drug resistant infections:
- A new podcast series, Superbugs and You, addresses the global AMR crisis through conversations with patients, clinicians, and researchers to discover how superbugs are affecting people and healthcare systems globally.15
- The recently released U.S. 2020-2025 National Action Plan accelerates response to antibiotic resistance by presenting coordinated, strategic actions to improve the health and well-being of all Americans across the One Health spectrum.16 One Health is a collaborative, multisectoral, and transdisciplinary approach – working at the local, regional, national, and global levels—with the goal of achieving optimal health outcomes recognizing the interconnection between people, animals, plants, and their shared environment.
- A new report from Wellcome Trust - The global response to AMR momentum, success, and critical gaps – was released in November 2020. This report gives an update on the status quo, recent developments and remaining critical gaps in the AMR response globally. One of the notable recent successes that was highlighted in the report was “the AMR community has grown into a broad, multi-sectoral coalition of actors that come from a range of sectors, including human health, animals and agriculture, and the environment.”17
Surveillance, infection prevention, and diagnostic stewardship have been deployed successfully to combat the global COVID-19 pandemic. Continued focus on these practices with improved antibiotic stewardship efforts need to be deployed to combat the insidious threat of drug resistant infections.
References:
- Antimicrobial resistance. World Health Organization. https://www.who.int/news-room/fact-sheets/detail/antimicrobial-resistance#:~:text=Antimicrobial%20resistance%20(AMR)%20is%20a,public%20health%20threats%20facing%20humanity. Accessed March 1, 2021.
- No time to wait: securing the future from drug-resistant infections. World Health Organization. https://www.who.int/antimicrobial-resistance/interagency-coordination-group/final-report/en/. Accessed March 28, 2021.
- Talkington K. Superbugs don’t respect borders. October 2017. The Pew Charitable Trusts. https://www.pewtrusts.org/en/research-and-analysis/articles/2017/10/10/superbugs-dont-respect-borders.Accessed February 25, 2021.
- Candida auris. Centers for Disease Control and Prevention. https://www.cdc.gov/fungal/candida-auris/index.html. Accessed February 26, 2021.
- Chen J, Tian S, Han X, et al. Is the superbug fungus really so scary? A systematic review and meta-analysis of global epidemiology and mortality of Candida auris. BMC Infect Dis. 2020;20(1):827. doi:10.1186/s12879-020-05543-0.
- Hall, W, McDonnell, A, O’Neill J. Superbugs: An Arms Race Against Bacteria. Cambridge, MA: Harvard University Press; 2018.
- Wee LEI, Conceicao EP, Tan JY, et al. Unintended consequences of infection prevention and control measures during COVID-19 pandemic [published online ahead of print, 2020 Nov 4]. Am J Infect Control. 2020; S0196-6553(20)30963-9. doi:10.1016/j.ajic.2020.10.019.
- Bentivegna E, Luciani M, Arcari L, Santino I, Simmaco M, Martelletti P. Reduction of multidrug-resistant (MDR) bacterial infections during the COVID-19 pandemic: a retrospective study. Int J Environ Res Public Health. 2021;18(3):1003. doi:10.3390/ijerph18031003.
- Vaccines to tackle drug-resistant infections: an evaluation of R & D opportunities. Wellcome Trust, https://vaccinesforamr.org/. Accessed March 28, 2021.
- Mahrous AJ, Thabit Pharm DAK, Elarabi S, Fleisher J. Clinical impact of pharmacist-directed antimicrobial stewardship guidance following blood culture rapid diagnostic testing. J Hosp Infect. 2020; 106 (In Press): 436-446. doi.org/10.1016/j.jhin.2020.09.010.
- Claeys K, Heil E, Hitchcock S, Johnson J, Leekha S, Management of gram-negative bloodstream infections in the era of rapid diagnostic testing: impact with and without antibiotic stewardship. Open Forum Infectious Diseases. 2020; 7(10): ofaa427. https://doi.org/10.1093/ofid/ofaa427.
- Bianco G, Boattini M, Iannaccone M, Sidoti F, Cavallo R, Costa C. Detection of antibiotic resistance genes from blood cultures: performance assessment and potential impact on antibiotic therapy management. J Hosp Infect. 2019; 102: 465-469. doi.org/10.1016/j.jhin.2019.03.007.
- Magill SS, O’Leary E, Ray SM, et al. Assessment of the appropriateness of antimicrobial use in U.S. hospitals. JAMA Netw Open. 2021;4(3):e212007. doi:10.1001/jamanetworkopen.2021.2007.
- Stewart Williams J, Wall S. The AMR emergency: multi-sector collaboration and collective global policy action is needed now. Glob Health Action. 2019;12(sup1):1855831. doi:10.1080/16549716.2019.1855831.
- Superbugs and you: true stories from scientists and patients around the world. Antimicrobial Resistance Fighter Coalition. 2020. https://antimicrobialresistancefighters.org/podcasts. Accessed March 25, 2021.
- U.S. national action plan for combating antimicrobial-resistant bacteria (national action plan). Centers for Disease Control and Prevention. 2020. https://www.cdc.gov/drugresistance/us-activities/national-action-plan.html. Accessed March 25, 2021.
- The Global response to AMR momentum, success, and critical gaps. Wellcome Trust. November 2020. https://cdn.eventsforce.net/files/ef-lpifs4q56r2a/website/785/wellcome-global-response-amr-report.pdf. Accessed March 29, 2021.
About the Author

Diane Flayhart, MBA
is the Global Program Leader for Antimicrobial Resistance at BD. Flayhart works to champion and support development of new Antimicrobial Resistance (AMR) program initiatives in global awareness, infection prevention/control and antimicrobial stewardship. Flayhart leads efforts for the Antimicrobial Resistance Fighter Coalition, which is mobilized by BD.