The “Goldilocks Approach” to molecular diagnostics stewardship
When it comes to patient care, I think it is fair to say that clinicians, lab professionals — all of us who work in healthcare — want to get it 'just right' for every patient, starting at diagnosis.
Correct and timely diagnosis is essential for effective treatments and improving patient outcomes.1 In modern medicine, diagnostic tests figure prominently in clinical decision making surrounding a new diagnosis.
In infectious disease care, we've benefited from decades of technological advances designed to get to a diagnosis efficiently and cost effectively. Increasingly, however, advanced technologies introduce a new level of complexity and variety of options that must be managed if we are to get to 'just right,' right away.
Navigating the contribution–complexity paradox
For example, nucleic acid amplification-based tests for the diagnosis of infectious diseases, including polymerase chain reaction (PCR) tests, have rapidly expanded over the past two decades. With the increased availability and complexity of these tests, there is also an increased need for collaborative approaches to optimize test use to promote positive impacts on patient care, while mitigating potential negative impact or resource waste.2
Multiplex PCR tests (also known as 'syndromic panels') offer a timely example of this contribution-complexity paradox. Syndromic panels combine tests for numerous pathogens and resistance genes into a single test, and have offered a comprehensive approach to diagnosing infections, potentially leading to improved patient care and clinical workflow.3
Over the course of a few years, we've seen syndromic panel testing expand to multiple commercial assays for detection of respiratory, blood, gastrointestinal (GI), acute meningitis and encephalitis (ME), and lower respiratory tract infections (LRTIs).4 These panels are now fully integrated into many clinical laboratories' standard testing practices,4 bringing institutions potential benefits such as increasing diagnostic yield and helping clinicians identify co-infections.
Yet, questions persist regarding syndromic panels, notes a 2023 joint society report published in the Journal of Microbiology.5 While multiplex diagnostic approaches offer clear benefits, the report emphasizes that several critical uncertainties remain. These include determining the optimal breadth of panel targets, evaluating whether broad multitarget panels outperform more selective testing strategies, and assessing whether faster results alone drive improved outcomes — or if a more algorithmic, cost-conscious approach could better serve patients. As clinical evidence continues to evolve, these questions underscore the need for ongoing research and thoughtful implementation.
Antigen testing is another example of a contribution–complexity trade-off. These tests offer rapid results — often within minutes — which can be critical for timely decision-making, especially during outbreaks or in resource-limited settings. However, this speed comes at a cost: antigen tests are generally less sensitive than molecular tests like PCR, meaning they may miss infections, particularly early or late in the course of illness. For example, studies found that antigen test sensitivity ranged from 47% to 64% when compared with PCR.6
This trade-off between speed and accuracy illustrates the complexity of choosing the right diagnostic tool for the right situation. As highlighted in JAMA and Centers for Disease Control and Prevention (CDC) reports,7,8 while antigen tests can help identify infectious individuals quickly, they may not be sufficient for high-risk patients who require more accurate diagnosis for treatment decisions. In addition, the potential for false negative results due to the lower sensitivity of antigen tests may increase the risk for secondary cases and hospital-acquired respiratory infections.9
Fortunately, the conversation regarding how, when, and why to deploy different tests dovetails with calls for widespread adoption of diagnostic stewardship, which is the practice of ordering the right tests for the right patient at the right time to inform optimal clinical care.10
Making diagnostic stewardship part of everyday healthcare
Diagnostic stewardship aims to improve the entire diagnostic process — from test ordering to performance to result reporting — by ensuring tests are used appropriately, collected correctly, and understood by both clinicians and patients.11 It has long been a topic of conversation among laboratorians, clinicians, and other healthcare providers who prioritize patient-centered care. Now, however, there is increased attention on the topic and a sense of urgency for providers to codify and adopt diagnostic stewardship programs across the care continuum.
This urgency is in part driven by two landmark reports in 2024 and 2025 from the CDC,1 developed with input from a broad array of stakeholders. Notably, the 2025 report — Core Elements of Hospital Diagnostic Excellence — defines six Core Elements for providers to adopt and adapt to their institutions. 'Promote diagnostic stewardship is featured in the core element 'Action,' and the report includes guidance on operationalizing stewardship practices as part of a hospital-based Diagnostic Excellence program.
I applaud this ground-breaking work, which shies away from any notion that there is a 'one-size fits all' stewardship prescription. I wholeheartedly concur that what is called for are ways to help providers find their 'just right' program. I propose The Goldilocks Approach offers a way forward.
The Goldilocks Approach to molecular diagnostics stewardship
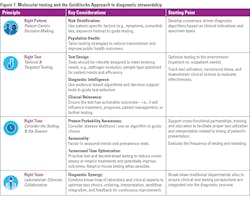
The Goldilocks Approach is a patient-centered, evidence-based set of principles and practices intended to help providers realize diagnostic stewardship across the care continuum and for all health conditions. Rooted in four principles: Right Patient, Right Test, Right Time, and Right Team (See Figure 1), the Goldilocks Approach works synergistically with the CDC recommendations with prompts to help providers formulate and activate a stewardship program that aligns to their resources and resolve.
Further, the Goldilocks Approach was developed with real-world contribution–complexities top of mind. For example, returning to the molecular diagnostics examples cited earlier, introduction of a Goldilocks Approach will prompt thoughtful dialogue regarding stewardship of syndromic panels (is more always better?) and antigen testing (what are our acceptable speed/accuracy trade-offs per patient cohort?), among other topics.
Crossing the chasm together
We're at an exciting time in the evolution of molecular diagnostics, propelled forward by technological advances that offer proven benefits and introduce complexity in clinical decision-making. A scientific paper from Zanella et al. frames the challenge clearly: "Clinicians are facing a choice: should testing drive clinical management or should medical needs stay in the driver seat, with decisions to test and treat target(s) remaining secondary to a stepwise clinical strategy that includes consideration of epidemiological factors."12 This tension reflects a broader shift in healthcare — one that calls for diagnostic stewardship, where testing supports, rather than dictates, clinical decision-making. The goal is not just more testing, but smarter testing, aligned with patient needs and public health priorities.
I anticipate ample and robust discussion of diagnostic stewardship will continue. It is my intent, and my hope, that the Goldilocks Approach contributes to that dialogue and catalyzes widespread adoption of common sense practices that help providers get care 'just right' for every patient.
References
- Core elements of hospital diagnostic excellence (DxEx). Centers for Disease Control and Prevention. Published February 11, 2025. Accessed January 29, 2026. https://www.cdc.gov/patient-safety/hcp/hospital-dx-excellence/index.html.
- Valencia-Shelton F, Anderson N, Palavecino EL, et al. Approaches to developing and implementing a molecular diagnostics stewardship program for infectious diseases: an ASM Laboratory Practices Subcommittee report. J Clin Microbiol. 2024;62:e00941-24. doi:10.1128/jcm.00941-24.
- Dumkow LE, Worden LJ, Rao SN. Syndromic diagnostic testing: a new way to approach patient care in the treatment of infectious diseases. J Antimicrob Chemother. 2021;76(Suppl 3):iii4-iii11. doi:10.1093/jac/dkab245.
- Dien Bard J, McElvania E. Panels and syndromic testing in clinical microbiology. Clin Lab Med. 2020;40(4):393-420. doi:10.1016/j.cll.2020.08.001.
- Lewinski MA, Alby K, Babady NE, et al. Exploring the utility of multiplex infectious disease panel testing for diagnosis of infection in different body sites: A joint report of the Association for Molecular Pathology, American Society for Microbiology, Infectious Diseases Society of America, and Pan American Society for Clinical Virology. J Mol Diagn. 2023;25(12):857-875. doi:10.1016/j.jmoldx.2023.08.005. Erratum in: J Mol Diagn. 2025;27(3):232. doi:10.1016/j.jmoldx.2024.12.004.
- Smith-Jeffcoat SE, Mellis AM, Grijalva CG, et al. SARS-CoV-2 viral shedding and rapid antigen test performance - Respiratory Virus Transmission Network, November 2022-May 2023. MMWR Morb Mortal Wkly Rep. 2024;73:365-371. doi:10.15585/mmwr.mm7316a2.
- Chu VT, Schwartz NG, Donnelly MAP, et al. Comparison of home antigen testing with RT-PCR and viral culture during the course of SARS-CoV-2 infection. JAMA Intern Med. 2022;182(7):701-709. doi:10.1001/jamainternmed.2022.1827.
- Kennedy AG. Evaluating the effectiveness of diagnostic tests. JAMA. 2022;327(14):1335-1336. doi:10.1001/jama.2022.4463.
- Ghazi L, Simonov M, Mansour SG, et al. Predicting patients with false negative SARS-CoV-2 testing at hospital admission: A retrospective multi-center study. PLoS One. 2021;16(5):e0251376. doi:10.1371/journal.pone.0251376.
- Fabre V, Davis A, Diekema DJ, et al. Principles of diagnostic stewardship: A practical guide from the Society for Healthcare Epidemiology of America Diagnostic Stewardship Task Force. Infect Control Hosp Epidemiol. 2023;44(2):178-185. doi:10.1017/ice.2023.5.
- Singh HK, Claeys KC, Advani SD, et al. Diagnostic stewardship to improve patient outcomes and healthcare-associated infection (HAI) metrics. Infect Control Hosp Epidemiol. 2024;45(4):405-411. doi:10.1017/ice.2023.284.
- Zanella MC, Meylan P, Kaiser L. Syndromic panels or “panel syndrome”? A perspective through the lens of respiratory tract infections. Clin Microbiol Infect. 2020;26(6):665-668. doi:10.1016/j.cmi.2019.12.018.
About the Author

Connie Savor, MD, MBA
serves as Chief Medical Officer for Cepheid, a molecular diagnostics company that makes the GeneXpert systems and Xpert tests.