The gold-standard
sample for blood-gas analysis is arterial blood obtained via an
indwelling arterial catheter or by arterial puncture. For a number of
reasons, capillary blood is an attractive substitute sample that is
routinely used in some clinical settings. The purpose of this article is
to examine the evidence that blood-gas parameter values (pH, pCO2,
and pO2) obtained from a capillary-blood sample
accurately reflect arterial blood. There is conflicting opinion that
increasing local blood flow (by warming or application of vasodilating
agent) prior to capillary-blood sampling is necessary for most accurate
results and this controversial issue will be addressed.
[Note: The unit of pCO2 and pO2 measurement
used in this article is kPa — to convert kPa to mmHg divide by 0.133.]
Blood-gas analyzers measure blood pH, and the oxygen
and carbon-dioxide tensions of blood (pCO2 and pO2).
These measurements, along with parameters (bicarbonate, base excess, and so
on) derived by calculation from these measurements, allow evaluation of
acid-base status and adequacy of ventilation and oxygenation. Thus,
blood-gas analysis is helpful for assessment and monitoring of patients
suffering a range of metabolic disturbances and respiratory diseases, both
acute and chronic. It is an important component of the physiological
monitoring that critically ill patients, particularly those being
mechanically ventilated, require.
The gold-standard sample for blood-gas analysis is
arterial blood obtained anaerobically via an indwelling arterial catheter
(most often sited at the radial artery in adults and the umbilical artery in
neonates), or arterial puncture. In an intensive-care setting where patients
may require frequent (perhaps two hourly) blood-gas testing, arterial
catheterization may be justified because it allows not only convenient and
painless access to arterial blood but also continuous blood-pressure
monitoring. Placing an arterial catheter is, however, an invasive, painful,
and technically difficult procedure,1 which is associated with
risk of serious complications including systemic infection, hemorrhage,
thrombosis, and ischemia.2 Technical and safety considerations
determine that, for most patients who require blood-gas analysis, placement
of an arterial catheter is either not justified or justified for only a
limited period, so that arterial blood is most often sampled by arterial
puncture using needle and syringe.
The most usual puncture site is the radial artery in
the wrist; alternative sites include the brachial artery in the arm and
femoral artery in the groin. Although arterial puncture does not place
patients at risk of the serious complications associated with arterial
catheterization, it is potentially hazardous and certainly not risk free.3
Furthermore, it is a procedure that is reported by patients to be
significantly more painful than venous puncture.4 Specialist
training in arterial puncture is essential for patient safety and comfort;
and, in many countries, obtaining arterial blood is the almost exclusive
preserve of medically qualified staff.
Capillary blood can be obtained by near-painless5
skin puncture using a lancet or automated incision device that punctures the
skin to a depth of just 1 millimeter.6,18 It is the
least-invasive and safest blood-collecting technique, and can be performed
by all healthcare personnel after minimal training.9 The relative
simplicity and safety profile of capillary-blood sampling and the necessity
for only small volumes (100 uL to 150 uL) of blood for pH and gas analysis
make capillary blood an attractive substitute for arterial blood,
particularly among neonates and infants but also adults. The clinical value
of capillary-blood gas results depends, however, on the extent to which pH,
pCO2, and pO2 of capillary blood
accurately reflect pH, pCO2, and pO2 of
arterial blood.
Capillary and arterial blood: theoretical considerations
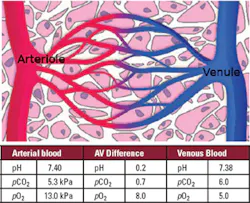
With a diameter of just 8 um, capillaries are the
smallest blood vessel. They are the connection between arterioles (the
smallest artery) and venules (the smallest vein) and, thus, between the
arterial and venous sides of the circulatory system. The capillary network
(see Figure 1) is the site of nutrient and waste exchange between blood and
tissue cells, made possible by the single-cell (1-um) thickness of the
capillary wall. Oxygenated arterial blood arriving via arterioles at the
capillary network yields up its oxygen and other essential nutrients to
tissue cells as carbon dioxide and other waste products of metabolism are
added to blood for transport from tissue cells via venules and the venous
system. As a consequence of these exchanges, there is a pH, pCO2,
and pO2 gradient across the capillary network (from
arteriole to venule), known as the arteriovenous (AV) difference. For
example, the pO2 of blood in arterioles is normally 13 kPa,
but, following loss of oxygen to tissues, is only 5 kPa in venules, giving
an AV difference for pO2 of approximately 8 kPa.7 The
normal AV differences for pH and pCO2 are of the order
0.02 pH to 0.03 pH units and 0.6 kPa to 0.7 kPa, respectively.8
Given the anatomical relationship of capillaries to
arterioles and venules, it might be supposed that the pH, pCO2,
and pO2 of capillary blood would lie roughly midway
between arterial and venous values. That is, however, not the case because
blood obtained by skin puncture is not actually pure capillary blood but a
mixture of blood from punctured arterioles, capillaries, and venules
(along with a small but variable contribution of interstitial fluid and
intracellular fluid from damaged tissue cells).9 Due to the
relative high pressure on the arterial side of the circulation, this blood
mixture contains a relatively greater proportion of blood from the arteriole
side of the capillary bed than from the venule side; and, thus, a
“capillary-blood” sample obtained by skin puncture approximates closer to
arterial blood than venous blood. This is the theoretical justification for
the use of capillary blood as a substitute for arterial blood.
AV difference is clearly a major theoretical
determinant of difference between arterial- and capillary-blood-gas values.
The greater the AV difference, the worse the agreement.10 By this
argument, it can be predicted that pO2 (which exhibits a
relatively high AV difference) is less likely to show good agreement between
capillary and arterial blood than pCO2 and pH (which both,
by comparison, have a low AV difference). Furthermore, reduced pO2
(hypoxemia) is associated with reduction in AV difference and hyperoxemia
with increased AV difference.7 There is good theoretical reason
to suppose that capillary- and arterial-blood pO2 will
agree more closely if arterial pO2 is reduced than if
arterial pO2 is normal or raised.
So long as tissue oxygen consumption and
carbon-dioxide production remains unchanged, as is the case in the resting
state, increasing blood flow through the capillary bed has the effect of
reducing AV difference and, thereby, the difference between arterial and
capillary pH, pCO2, and pO2. This
provides the rationale for strategies such as pre-warming the puncture site
or treating the puncture site with vasoactive agents prior to
capillary-blood sampling. The increased local blood flow that is presumed to
occur when these pre-sampling strategies are adopted — theoretically, at
least — leads to so called “arterialization” of capillary blood and pH, pCO2,
and pO2 values that more accurately reflect those of
arterial blood.
Arterial- and capillary-blood-gas pH,
pCO2, and pO2: study findings
Several studies of healthy individuals have defined
reference ranges for capillary blood pH, pCO2, and pO2.11,12
The results of at least one11 demonstrate that for healthy
adults, sampling “arterialized” capillary blood provides results that are
not significantly different from those obtained from arterial blood
(see Table 1). Of greater significance are the many more studies conducted
over the past 40 years10,13-27 that have compared blood-gas
values of simultaneously collected capillary and arterial blood in patients
whose clinical condition demands blood-gas analysis. In general, they have
revealed while capillary-blood pH and pCO2 reflects
arterial pH and pCO2 sufficiently accurate for clinical
purposes, that may not be the case for pO2. Studies in this area
have focused exclusively on either pediatric patients (mostly neonates)13-21
or adult patients.10,21-27
Studies of pediatric patients: The
capillary blood for all studies of neonates and young infants13-16,18,20
was sampled by heel stab. The method of arterialization was almost
exclusively heel warming, usually by immersing the heel in warm water (40^0C
to 45^0C) for five to 10 minutes prior to heel stab, or using a warmed
surgical-plastic device.18 The rather cumbersome method of
histamine iontophoresis was used to arterialize capillary blood of neonates
in one early study.15 Finger stab was the preferred site for
sampling capillary blood from children.17,19
The vast majority of studies reveal clinically
acceptable agreement between capillary and arterial pH — a difference of
less than 0.05 pH units being considered clinically insignificant.16,17
In one study19 of 75 paired samples, the mean of capillary-pH
results was identical to the mean of arterial results; and, in all other
studies, the mean difference ranged from 0.001 pH units14 to 0.02
pH units.20 One of the larger studies in which 158 paired samples
from 41 pre-term neonates were compared,16 despite a mean
difference of just 0.001 pH units, 24% of paired samples gave clinically
discrepant results (i.e., a difference of >0.05 pH units). This, however,
did not detract the authors from the conclusion that capillary blood is a
“satisfactory” alternative to arterial blood for measurement of pH. Closer
agreement was revealed by a later study17 of 50 babies and
children being cared for in a pediatric intensive-care unit. Here, the mean
difference between capillary and arterial pH was just 0.009 pH units (95%
limits of agreement +/-0.032) and in no patient was there a difference greater
than 0.05 pH units. Johnson, et al,8 and Hunt15
reported no significant difference between capillary and arterial pH of 21
sick neonates (aged from three hours to seven days) and 44 sick babies (aged
3.5 days to 10 weeks).
In common with pH, most studies reveal clinically
acceptable agreement between “arterialized” capillary pCO2
and arterial pCO2 — a difference of less than 1 kPa16
or less than 0.87 kPa17 being considered clinically
insignificant. All studies revealed the same bias with mean of capillary
pCO2 values greater than the mean of arterial pCO2,
although this difference was in most studies small — ranging from 0.04 kPa20
to 0.21 kPa.17 This last study17 showed clinically
discrepant results (difference greater than 0.87 kPa) in only two out of 50
(4%) paired samples. One of few studies to have revealed poor agreement was
that of Hunt.15 Here, the mean difference between capillary and
arterial pCO2 was 2.0 kPa.
All studies reveal a bias with regard to pO2
with mean of arterial pO2 values greater than mean of
capillary pO2 values. Most studies reveal that this
difference is of sufficient magnitude to conclude that there is an
unacceptably low level of agreement between capillary and arterial pO2.
McClain, et al,16 found a mean pO2 difference of 2.4
kPa, with 84% of paired samples differing by more than 1 kPa, 56% differing
by more than 2 kPa, and 24% differing by more than 3 kPa. They judge that
difference of >1 kPa has clinical significance. Harrison, et al,17
found a mean difference in pO2 between the sample types of
3.3 kPa. In 42 of 50 (84%) paired samples, the difference exceeded 0.87 kPa
— the difference limit they had set for clinical acceptability. Bland-Altman
plot revealed that the magnitude of the difference between arterial and
capillary pO2 depends on arterial pO2.
As arterial pO2 increases so, too, does the difference
between capillary and arterial pO2. Conversely, as
arterial pO2 decreases, the difference decreases. So
striking was this effect that Harrison, et al, found acceptable agreement
between capillary and arterial pO2 in all paired samples
with arterial pO2 less than <8 kPa. This is in agreement
with other studies that have included sufficient numbers of severely
hypoxemic neonates.16,15
acceptable sample alternative to arterial blood if only acid-base
parameters (pH and pCO2) are of interest.
Arterialization strategies not effective:
Several of these studies15,16,18 tested the effectiveness of
strategies aimed at arterializing capillary blood. Hunt, et al,15
simultaneously collected arterialized (by histamine iontophoresis) and
non-arterialized capillary samples from each study subject for comparison
with arterial blood and found no difference in pH, pCO2,
and pO2 results for arterialized and non-arterialized
capillary samples. Likewise, Johnson, et al,18 found that warming
babies' heels in a plastic-molded heating device for, on average, seven
minutes had no arterializing effect; there was no significant difference in
pH, pCO2, and pO2 for capillary blood
sampled from a warmed heel compared with capillary blood sampled at the same
time from the contralateral unwarmed heel. The greatest arterializing effect
of heel warming was found by McLain, et al16; but, although mean
values for pH, pCO2, and pO2 of warmed
heel blood were all slightly closer to mean values for arterial blood than
were mean values derived from unwarmed heel blood, on statistical analysis,
the differences were again found to be insignificant. These results are in
accord with other recent studies28,29 that have found heel
warming has no effect in terms of improved blood flow, an indication of
effective arterialization.
A study conducted nearly 50 years ago30
provides limited evidence that heel warming is effective. This compared
capillary with arterial blood pH and pCO2 (but not pO2)
in 106 neonates (all less than two weeks old). In total, 149 sample pairs
were obtained for comparison. The heel was warmed before collection of
capillary blood in 126 instances. For the remaining 23 pairs, capillary
blood was collected without prior heel warming. Superior agreement was
observed for both pH and pCO2 for the 126 arterial vs.
warmed capillary-blood pairs compared with the 23 arterial vs. unwarmed
capillary-blood pairs.
The Clinical and Laboratory Standards Institute
(CLSI) document H4-A5 on the subject of capillary-blood sampling states:
“The need for heel warming is not universal in the literature. The cited
references provide data showing no significant difference of analyte
measurement (pH, blood gas, electrolytes) between warming and non-warming
for capillary collection. Although studies show that pre-warming may not be
necessary when using a skin-incision device, increasing blood flow may be
necessary to prevent hemolysis and/or contamination with tissue fluids when
using other devices or as a general practice.”
for arterial
and “arterialized” capillary blood
Studies of adult patients: The fingertip or, more commonly, the
lower tip of the earlobe are the usual sites of capillary-blood sampling in
adults, and the most common method of arterialization is application of a
vasodilating cream (e.g., Algipan) to the puncture site five to 10 minutes
prior to blood sampling. Of many studies22-27 that have compared
“arterialized” capillary-blood gases with arterial-blood gases in adults,
probably the most informative is a recently published meta-analysis by
Gerald Zavorsky and colleagues at McGill University.27 The
database that this group recovered from 29 previously published studies
comprised 664 paired samples for comparison of earlobe capillary with
arterial blood and 222 paired samples for comparison of fingertip-capillary
blood with arterial blood. The pH of these 886 paired samples ranged from
6.77 to 7.74; pCO2 from 1.3 kPa to 15.1 kPa, and pO2
from 2.8 kPa to 20.6 kPa.
Both fingertip- and earlobe-capillary pH were found
to accurately reflect arterial pH. The mean difference between arterial- and
earlobe-capillary pH was 0.01 +/- 0.02 pH units. Regression analysis for this
comparison revealed a coefficient of determination (r2) = 0.94
and a residual standard error of 0.025. Very similar results were found for
analysis of fingertip capillary vs. arterial pairs.
Capillary pCO2 values were also
found to be very close to those of arterial blood.
Mean difference between arterial- and
earlobe-capillary blood pCO2 was 0.01 kPa +/- 0.38.
Coefficient of determination (r2) = 0.94; residual standard error 0.4 kPa.
Although judged acceptable, agreement between fingertip-capillary and
arterial pCO2
was not as close as that between earlobe-capillary and arterial pCO2.
There was poor agreement between fingertip-capillary
and arterial pO2. Mean difference was 1.4 +/- 2.0 kPa.
Coefficient of determination (r2) = 0.48; residual standard error 2.0 kPa.
By comparison, better agreement was evident between earlobe-capillary and
arterial pO2. Here, mean difference was 0.3 +/- 0.8 kPa.
Correlation of determination (r2) = 0.88; residual standard error 0.8 kPa.
There was unequivocal evidence that agreement between capillary (both types)
and arterial pO2 improves as arterial pO2
falls.
The authors of this significant study conclude that
capillary blood sampled from either the fingertip or earlobe (preferably),
accurately reflects arterial pH and pCO2 over a wide range
of values. Sampling blood from the earlobe (but never the fingertip) may be
an appropriate substitute for arterial pO2 unless
precision is required. The large standard error associated with
earlobe-capillary-pO2 measurement limits its clinical
usefulness.
There is consensus that capillary blood is a
clinically acceptable sample alternative to arterial blood if only acid-base
parameters (pH and pCO2) are of interest. Most studies
conducted prior to the mid-1990s5,13,22,23 suggested that
capillary-blood
pO2 reflected arterial pO2 sufficiently
accurately for clinical purposes and that capillary blood could justifiably
be used as a substitute for arterial blood, not only to assess patient
acid-base status but also oxygenation status. The results of recent studies10,18-21,24-27
have challenged that view; and the relatively poor agreement between
capillary and arterial pO2 — most marked if pO2
is raised and least marked if pO2 is low — revealed by
these studies, suggest that capillary-pO2 results have
limited clinical value and should be interpreted with caution. Capillary
blood sampled from the fingertip is particularly unsuited for assessment of
oxygenation status.27 There is really no substitute for arterial
blood if accuracy of pO2 measurement is important, for
example, for the prescription of long-term oxygen therapy.26
There is evidence from several studies to suggest that the ritual of warming
the heel of babies prior to sampling capillary blood is not effective in
“arterializing” capillary blood. There is little, if any, contrary evidence
to suggest it is effective. The effectiveness of vasodilating agents
in “arterializing” earlobe-capillary blood samples seems not to have been
formally assessed.
Freelance writer Chris Higgins is a regular
contributor to bloodgas.org, which provides practical information to help
healthcare professionals deal with daily issues surrounding blood-gas and
acute-care testing. His master's degree is in medical biochemistry, and of
his 20 years' work experience in clinical laboratories, 10 were as a
manager.
Editor's note:
Reprinted with permission from
bloodgas.org, a knowledge website sponsored and maintained by
Radiometer. On bloodgas.org, international experts and healthcare
professionals share their knowledge and real-life experiences, helping to
ensure the quality, credibility, and relevance of the information found on
the site. Radiometer develops and produces advanced blood-gas analyzers and
other medical instruments that quickly provide accurate information about
the condition of critically ill patients. Radiometer gives special attention
to providing users with the skills and knowledge they need to get the most
out of their solutions. Sponsoring and maintaining
bloodgas.org is, therefore, a natural extension of the company's
existing knowledge activities.
References
1. Eisen L, Miami T, Berger J et al.
Gender disparity in failure rate for arterial catheter attempts. J
Intensive Care. 2007;22:166-172.
2. Wallach SG. Cannulation injury of the radial
artery: diagnosis and treatment algorithm. Am J Crit Care.
2004;13:315-319.
3. Okeson G, Wullbrecht P. The safety of brachial
artery puncture for arterial blood sampling. Chest.
1998;114:748-751.
4. Giner J, Casan P, Belda J, et al. Pain during
arterial puncture. Chest. 1996;110:1443-1445.
5. Dar K, Williams T, Aitken R, et al. Arterial
versus capillary sampling for analysing blood gas pressures. BMJ.
1995;310:24-25
6. Paes B, Janes M, Vegh P, et al. A comparative
study of heel stick devices for infant blood collection. Am J Dis
Child. 1993;147:346-348.
7. Hughes J. Blood gas estimations from
arterialized capillary blood versus arterial puncture: are they
different? Eur Respir J. 1996;9:184-185.
8. Toftegaard M, Rees S, Andreeason S.
Correlation between acid-base parameters measured in arterial blood and
venous blood sampled peripherally, from the vena cava and from the
pulmonary artery.
Eur J Emerg Med. 2008;15:86-91.
9. Burnett RW, Covington AK, Fogh-Anderson N, et
al. Approved IFCC recommendations on whole blood sampling, transport and
storage for simultaneous determination of pH blood gases and
electrolytes. Eur J Chem Clin Biochem. 1995;33:247-253.
10. Sauty A, Uldry C, Debatez L-F. Differences in
pO2 and pCO2 between arterial and arterialised earlobe
samples. Eur Respir J. 1996;9:186–189.
11. Dong SH, Liu HM, Song GW, et al. Arterialised
capillary blood gases from acid-base studies in normal individuals from
29 days to 24 years of age. Am J Dis Child. 1985;139:1019-1022.
12. Cousineau J, Anctil S, Carcellar A, et al.
Neonatal capillary blood gas reference values. Clin Biochem.
2005;38:905-907.
13. MacRae DJ, Palavradi D. Comparison between
arterial, capillary and venous acid-base measurements in the newborn
infant. J Obstet Gynae of Br Commonwealth. 1966;73:761-765.
14. Desai S, Holloway R, Thambiran A, et al. A
comparison between arterial and arterialised capillary blood in infants.
S Afr Med J. 1967;41:13-15.
15. Hunt C. Capillary blood sampling in the
infant: usefulness and limitations of two methods of sampling compared
with arterial blood. Pediatrics. 1973;51:501-506.
16. McLain B, Evans J, Dear P. Comparison of
capillary and arterial blood gas measurements in neonates. Arch Dis
Child. 1988;63:743-747.
17. Harrison A, Lynch J, Dean J. Comparison of
simultaneously obtained arterial and capillary blood gases in pediatric
intensive care patients. Crit Care Med. 1997;25:1904-1908.
18. Johnson K, Cress G, Connolly N et al.
Neonatal Laboratory blood sampling: comparison of results from arterial
catheters with those from an automated capillary device. Neonatal
Network. 2000;19:27–4.
19. Escalante-Kanashiro R, Tantalean-Da-Fieno J.
Capillary blood gases in a pediatric intensive care unit. Crit Care
Med.2000; 28: 224-226.
20. Yang K, SU B-H, Tsai F-J, Peng C-T. The
comparison between capillary blood sampling and arterial blood sampling
in an NICU. Acta Paediatr Taiwan. 2002;43:124-126.
21. Yildizas D, Yapicioglu H, Yilmaz H et al.
Correlation of simultaneously obtained capillary, venous and arterial
blood gases of patients in a paediatric intensive care unit. Arch Dis
Child. 2004;89:176-180.
22. Pitkin A, Roberts C, Wedzicha J. Arterialised
earlobe blood gas analysis: an underused technique. Thorax.
1994;49:364-366.
23. Dar K, Williams T, Aitlen R. Arterial versus
capillary sampling for analysing blood gas pressures. BMJ.
1995;310:24-25.
24. Dall'Ava-Santucci J, Dessanges J, Dinh Xaun A
et al. Is arterialised earlobe blood pO2 an acceptable
substitute for arterial blood pO2? Eur Respir J.
1996;9:1329-1330.
25. Fajac I, Texereau V, Rivoal J-F et al. Blood
gas measurement during exercise: a comparative study between
arterialized earlobe sampling and direct arterial puncture in adults.
Eur Respir J. 1998;11:712-715.
26. Eaton T, Rudkin S, Garrett J. The clinical
utility of arterialised earlobe capillary blood in the assessment of
patients for long term oxygen therapy. Respir Med.
2001;95:655-660.
27. Zavorsky G, Cao J, Mayo N. Arterial versus
capillary blood gases: a meta- analysis. Respir Physiol &
Neurobiology. 2007;155:268-279.
28. Janes M, Pinelli J, Landry S et al.
Comparison of capillary blood sampling using an automated incision
device with and without warming the heel. J Perinatology.
2002;22:154-158.
29. Barker DP, Willets B, Cappendijik V, et al.
Capillary blood sampling: should the heels be warmed? Arch Dis Child
(Fetal & Neonatal). 1996;74:F139-140.
30. Gandy G, Grann L, Cunningham N et al. The
validity of pH and pCO2 measurements in capillary samples in sick and
healthy infants. Pediatrics. 1964;34:192-197.