Respiratory illnesses: A growing opportunity for stewardship
To take the test online go HERE. For more information, visit the Continuing Education tab.
LEARNING OBJECTIVES
Upon completion of this article, the reader will be able to:
1. Discuss the challenges that the healthcare industry faces with respiratory illness diagnosis.
2. Define syndromic testing and describe its benefits.
3. Describe the benefits of multiplex PCR testing and its utility in diagnosing respiratory illnesses.
4. Describe how diagnostic and antimicrobial stewardship work to improve antimicrobial resistance.
Only the rare individual hasn’t been sickened by a respiratory infection — either viral or bacterial. After all, these pathogens are among the most common causes of disease in human beings,1 and a leading cause of death.2 Properly diagnosing respiratory pathogens can be extremely difficult to do in the observational physician office setting, primarily because the symptoms of influenza, respiratory syncytial virus (RSV), severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2), and other respiratory infections are often indistinguishable.3 Further complicating matters, the pandemic emergency caused shifting of assumed seasonal patterns, which have served as a “guide” for physicians’ observational diagnoses.
Pandemic emergency measures such as social distancing and masking disrupted typical seasonal patterns of respiratory infections, including muting influenza for two years. When these measures ended and borders reopened, viral pathogens began to circulate again, but not on their usual schedule. A study of the 2022 flu season in Australia found that the incidence of flu peaked fully two months earlier than it had for the entire decade prior — in mid-June versus mid-August. Given that Australians typically receive their seasonal flu vaccine between March and May, the early start to the flu season meant that a smaller proportion of the population had received their annual flu shot.4 The overlap of respiratory symptoms, along with the blurring of seasonal lines, reinforces the need for labs, and the doctors and public health officials who rely on them, to think in terms of syndromic diagnostics to help effectively manage patient and population health and monitor viral co-circulation.5
The COVID-19 pandemic accelerated the introduction of new methods to diagnose and treat viruses, and greatly heightened awareness of diagnostic stewardship programs. It also highlighted the clinical utility of multiplex polymerase chain reaction (PCR) assays, or syndromic diagnostic testing for infectious diseases. The ability to deliver a rapid and accurate diagnosis has significantly changed the way infectious disease clinicians and laboratorians manage patients and optimize workflow. Accurate, earlier diagnoses also enable more precise therapeutic decisions and infectious control measures.6
Syndromic testing
A syndrome is a set of symptoms and signs that are correlated with each other and often with a specific disease. For example, a respiratory syndrome may include symptoms or signs such as fever, cough, muscle aches, elevated heart rate, or abnormal laboratory results (e.g., white blood cell count). Patients may experience just a few of these symptoms which can make it challenging for a clinician to identify a causative pathogen based on non-specific symptoms alone.
In addition, a patient’s ability to recover from a respiratory infection varies widely.
Consider a patient with chronic obstructive pulmonary disease (COPD) or asthma, for example. These patients are more likely to become seriously ill from respiratory infections, as compared to a patient with no underlying lung or health conditions.7 These and other factors that impact patient outcomes include the type of pathogen, its resistance to treatment, the patient’s age and underlying health status, and importantly, the speed and accuracy of a diagnosis so proper treatment can begin promptly.
Misdiagnosis matters
At the point of care, physicians do not have the tools to differentiate among overlapping symptoms. In a study comparing the results of early identification in an office against the results of PCR and RNA sequencing of samples from the same patients, the study team found that skilled medical observation without laboratory testing identified only 37 percent of the study’s patients with respiratory tract infections.8 A brief retrospective analysis demonstrated that multiplex testing identified 59 percent positive targets and over 90 percent of co-infections not detected by low-plex testing.
Sensitive multiplex PCR panel testing is a fast and simple way to accurately diagnose patients when multiple respiratory pathogens are circulating. In fact, the Centers for Disease Control and Prevention and the Academy of Medical Sciences “strongly support multiplex testing” in this situation.9,10 Syndromic testing with multiplex molecular panels offers clinical labs a way to quickly distinguish between a broad array of respiratory pathogens. Unlike traditional diagnostic methods, such as bacterial culture and microscopy, PCR tests can test for a host of pathogens and deliver accurate results in about an hour.
Without question, the coronavirus disease pandemic brought the utility of multiplex PCR assays into sharp focus. By combining common pathogens capable of causing a specific syndrome into one panel, multiplex testing can reduce the time needed to provide a diagnosis and a precise therapeutic decision.11
Many clinical publications show that syndromic testing can support antimicrobial stewardship programs, improve patient outcomes, and reduce overall healthcare costs, lessening the impact of misdiagnosis.
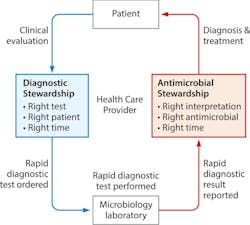
Diagnostic and antimicrobial stewardship
Many clinical publications show that syndromic testing can support antimicrobial stewardship programs, improve patient outcomes, and reduce overall healthcare costs, lessening the impact of misdiagnosis. Syndromic panels, if implemented thoughtfully and interpreted carefully, have the potential to improve antimicrobial use and patient outcomes through improved clinical decision making, optimized laboratory workflow, and enhanced antimicrobial and laboratory stewardship.12 Diagnostic stewardship refers to the appropriate use of laboratory testing to guide patient management, optimize patient outcomes, and limit the spread of antimicrobial resistance. Partnership among clinical laboratories, pharmacists and infectious disease clinicians can ensure that the right tests are ordered for the right patients, and the information they provide is translated into appropriate treatment decisions.13
In turn, appropriate treatment decisions can advance antimicrobial stewardship. Antimicrobial resistance is considered an urgent global health threat.14 In the United States, more than 2.8 million antimicrobial-resistant infections occur each year, and more than 35,000 people die as a result. The U.S. National Action Plan for Combating Antibiotic-Resistant Bacteria (CARB) includes a strategic goal to accelerate development and use of rapid and innovative diagnostic tests for identification and characterization of resistant bacteria.15 Syndromic testing can guide proper, evidence-based use of antibiotics16 — which do not work on viral infections — and support global Antimicrobial Stewardship Programs (ASPs).
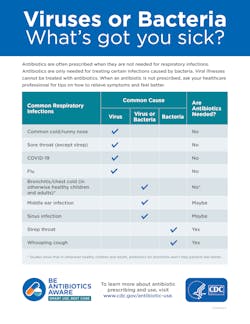
To mitigate the misuse of antibiotics for viral respiratory infections, the CDC provides education resources to help patients and clinicians recognize if their symptoms may be caused by a bacteria or virus (See Figure 1.)17
Additional benefits of syndromic testing to address respiratory infections
Syndromic testing is the subject of a growing number of studies, and has been shown to offer benefits to patients, reduce certain healthcare costs, and improve lab efficiency. Particularly for at-risk patients, speed matters. Laboratories can cement relationships with ordering physicians and care providers by offering the improved sensitivity and specificity of multiplex PCR testing and deliver accurate results in as little as an hour, instead of 2 or 3 days.18
Across several studies, use of syndromic testing to diagnose and guide treatment for respiratory infections led to a 13 percent reduction in antibiotic therapy duration,19 a 30 percent reduction in antibiotic prescriptions,20 and one (1.0) fewer days in median antimicrobial duration in adult influenza patients.21
Mortality and length of stay in intensive care units (ICU) are both reduced by syndromic testing, with one study3 reporting a three-day reduction in ICU days using multiplex syndromic tests as compared to batch testing and a 10 percent increase in survival when results were reported in less than seven hours.
Reinforcing the argument that quality, evidence-based care saves money, syndromic testing not only reduced ICU bed days, but delivered per patient ICU cost savings of over $9,000 with a positive respiratory pathogen and over $8,000 for a negative result.3
The benefits of syndromic testing for respiratory infections extends into the clinical laboratory, too. Among the significant benefits for labs, is the opportunity to streamline the workflow. One molecular test can provide multiple results so labs can reduce the number of low-plex, microscopy and culture tests ordered. Next, labs can optimize their operations. With less hands-on time and minimal training required, samples can be processed 24 hours a day, seven days a week, without waiting to batch samples. Also, labs were able to deliver a 30.4 hour reduction in turnaround time versus batch testing.22
Not all syndromic panels work the same way, but attractive features to look for include having all reagents preloaded into the cartridge, the ability to perform tests with a single-instrument solution, and cartridges that can be shipped and stored at room temperature.
Respiratory infections, caused by a wide range of pathogens, are likely to remain a significant source of illness among humans. As vaccines and treatments emerge to address viral respiratory pathogens, the role clinical laboratories can play is critical in differentiating what pathogen — bacterial or viral — has made a patient sick. Laboratories can drive the correct diagnosis and the correct treatment, meaning that diagnostic stewardship is a space that clinical laboratories are well positioned to define, underscoring the need for close partnership with front-line healthcare delivery providers, pharmacies, and public health agencies.
References
1. Infectious disease: Types, causes & treatments. Cleveland Clinic. Accessed July 19, 2023. https://my.clevelandclinic.org/health/diseases/17724-infectious-diseases.
2. The top 10 causes of death. Who.int. Accessed July 19, 2023. https://www.who.int/news-room/fact-sheets/detail/the-top-10-causes-of-death.
3. US Burden of Disease Collaborators, Mokdad AH, Ballestros K, et al. The state of US health, 1990-2016: Burden of diseases, injuries, and risk factors among US states. JAMA. 2018;319(14):1444-1472. doi:10.1001/jama.2018.0158.
4. Trent MJ, Moa A, MacIntyre CR. “I’ll be back”: Australia’s experience of flu in 2022. BMJ. 2022;379:o2998. doi:10.1136/bmj.o2998.
5. Chow EJ, Uyeki TM, Chu HY. The effects of the COVID-19 pandemic on community respiratory virus activity. Nat Rev Microbiol. 2023;21(3):195-210. doi:10.1038/s41579-022-00807-9.
6. Fox AS, Rao SN. Syndromic testing for the diagnosis of infectious diseases: the right test if used for the right patient. J Antimicrob Chemother. 2021;76(Suppl 3):iii2-iii3. doi:10.1093/jac/dkab248.
7. de Bellefonds C, Sinha S. COPD: Stay healthy during cold and flu season. Everydayhealth.com. Accessed July 19, 2023. https://www.everydayhealth.com/copd/stay-healthy-during-cold-flu-season/.
8. August. Study: Respiratory viruses easily misdiagnosed without better tests. Los Alamos National Laboratory. Accessed July 19, 2023. https://discover.lanl.gov/news/0831-respiratory-viruses/.
9. COVID-19: Preparing for the future. Acmedsci.ac.uk. Accessed July 19, 2023. https://acmedsci.ac.uk/file-download/4747802.
10. Testing guidance for clinicians when SARS-CoV-2 and influenza viruses are co-circulating. Cdc.gov. Published February 9, 2022. Accessed July 19, 2023. https://www.cdc.gov/flu/professionals/diagnosis/testing-guidance-for-clinicians-hospitaized.htm.
11. Fox AS, Rao SN. Syndromic testing for the diagnosis of infectious diseases: the right test if used for the right patient. J Antimicrob Chemother. 2021;76(Suppl 3):iii2-iii3. doi:10.1093/jac/dkab248.
12. Dumkow LE, Worden LJ, Rao SN. Syndromic diagnostic testing: a new way to approach patient care in the treatment of infectious diseases. J Antimicrob Chemother. 2021;76(Supplement_3):iii4-iii11. doi:10.1093/jac/dkab245.
13. Patel R, Fang FC. Diagnostic stewardship: Opportunity for a laboratory–infectious diseases partnership. Clin Infect Dis. 2018;67(5):799-801. doi:10.1093/cid/ciy077.
14. CDC. What Exactly is Antibiotic Resistance? Centers for Disease Control and Prevention. Published October 5, 2022. Accessed July 19, 2023. https://www.cdc.gov/drugresistance/about.html.
15. CDC. U.S. national Action Plan. Centers for Disease Control and Prevention. Published June 7, 2022. Accessed July 19, 2023. https://www.cdc.gov/drugresistance/us-activities/national-action-plan.html.
16. Benefits of syndromic testing. Qiagen.com. Accessed July 19, 2023. https://www.qiagen.com/us/applications/syndromic-testing/syndromic-testing-benefits.
17. Sick WGY. Viruses or Bacteria. Cdc.gov. Accessed July 19, 2023. https://www.cdc.gov/antibiotic-use/pdfs/VirusOrBacteria-Original-P.pdf.
18. Easy rapid molecular diagnostic tests. Qiagen.com. Accessed July 19, 2023. https://www.qiagen.com/us/applications/syndromic-testing/workflow.
19. Martinez, R.M, et al. Clinical Virology Symposium 2016. Poster #C-368
20. Zhu C, Sidiki S, Grider B, Fink B, Hubbard N, Mukundan D. A study of the use and outcomes from respiratory viral testing at a mid-sized children’s hospital. Clin Pediatr (Phila). 2019;58(2):185-190. doi:10.1177/0009922818809523.
21. Rappo U, Schuetz AN, Jenkins SG, et al. Impact of early detection of respiratory viruses by multiplex PCR assay on clinical outcomes in adult patients. J Clin Microbiol. 2016;54(8):2096-2103. doi:10.1128/jcm.00549-16.
22. US Burden of Disease Collaborators, Mokdad AH, Ballestros K, et al. The state of US health, 1990-2016: Burden of diseases, injuries, and risk factors among US states. JAMA. 2018;319(14):1444-1472. doi:10.1001/jama.2018.0158.
23. Messacar K, Parker SK, Todd JK, Dominguez SR. Implementation of rapid molecular infectious disease diagnostics: The role of diagnostic and antimicrobial stewardship.J Clin Microbiol. 2017;55(3):715-723. doi:10.1128/JCM.02264-16.About the Author

Anooj Shah, PharmD, MBA, BCIDP
is a Medical Science Liaison – Infectious Diseases for QIAGEN North America. A 2018 graduate of Concordia University, Wisconsin, Shah holds his Doctor of Pharmacy and his MBA. He completed two years of post-graduate residency training at Northwestern Medicine in Chicago, specializing in infectious diseases. Prior to joining QIAGEN in 2022, Shah served as a clinical pharmacist focusing on infectious diseases and antimicrobial stewardship.