When accuracy removes obstacles: The case for interference-free glucose meters
Over 500 million blood glucose tests are performed in U.S. hospitals every year.1 With the majority of these being done at the point-of-care (POC), accuracy of the devices used is paramount to guarantee the reliability of results and ensure patient safety. A common source of inaccuracy of blood glucose meters is interference from the myriad of medications or supplements that may be administered to support patients.
This is an even bigger issue in critically ill patients, where inaccurate glucose results can lead to devastating consequences for the patient.
Hospitals catering to this fragile patient population are therefore required to provide safe and effective care by equipping their staff with devices that are proven to mitigate these sources of inaccuracy. In this article, I discuss a case example of a hospital system that changed glucometers based on these considerations.
Policy and workflow burdens created by meter limitations
One of the main drivers for change of glucose meters came from the limitation of the current vendor whose meter was unable to correct for interfering substances such as ascorbic acid, N-acetylcysteine, and hematocrit. Because of the dangerous potential outcomes that have been reported with such interferences,2-5 our hospital system implemented policies and procedures to avoid using these meters on critically ill patients (see Figure 1). Unfortunately, these policies became cumbersome, confusing, and time consuming. With a multi-hospital healthcare organization, the various institutions and different levels of care amplified the complexity and burden of these policies. Our organization consists of three full-service hospitals, one emergency-only facility, and one long-term care facility. The main hub institution is a trauma center with all levels of intensive care from neonates to adults. Within the other two hospitals, there are critical care units, acute care units, and pediatric units. The emergency-only facility does not have any inpatient testing and has minimal laboratory testing. The long-term care facility also functions as a specialty hospital with no on-site laboratory testing. Across the organization, there are approximately 4,000 clinical staff certified to perform testing in the POC setting. While the POC program is expansive, the most utilized test is the glucometer. Transitioning to a new glucose meter for such a large number of staff was a large undertaking but was eased substantially with the support of the manufacturer who provided multiple training and support sessions.
Since the previous instrument was unsuitable for critical care patients, a policy was developed to restrict glucometer use in these populations (Figure 1). The policy aimed to limit the potential for false readings on the glucometer. However, the POC staff found it to be extremely challenging to enforce the policy or ensure that the providers and clinical staff were utilizing the instrument properly. If the clinical staff failed to use the policy correctly and the glucometer was used outside of the scope of service, it would push the meter from a CLIA waived test to a laboratory developed test (LDT) and potentially jeopardize patient care. LDTs are subject to significant regulatory restrictions including ongoing, time-consuming validations and caused the organization to re-evaluate their current instrumentation. Patient care would be impacted if these meters were used inappropriately on the wrong patient population since interferences can give rise to erroneously high results that could lead to fatal consequences of excessive insulin dosing, as described at other institutions.2-5
To eliminate the cumbersome policies, there was a need for a meter FDA cleared for critically ill patients. After evaluating all options, we chose a new glucose meter that was able to correct for the interferences and was FDA cleared for critically ill patients — enabling streamlined workflows, improved clinical accuracy, reduced policy complexity, and enhanced staff efficiency.
Transition process
The transition occurred over several months and included organization-wide training, validation, and implementation. The manufacturer initiated rolling education sessions for staff prior to going live. We started with the smallest hospital to learn best practices and moved progressively larger. The manufacturer validated all meters at one time followed by week-long training at facility 1. During this time, supplies that were necessary for facility 2 were ordered. Training sessions included “train-the-trainer” at the larger sites. There was never more than one hospital per week going live. This allowed time for meetings to discuss the lessons learned that could be applied at the next site. Overall, a progressive go-live with extensive validation and training support from the manufacturer made this an efficient and productive experience.
Technology change boosts POC testing efficiency and confidence
After implementation, we observed several significant improvements to the POC program. As anticipated, we saw improved efficiencies by elimination of the critically ill policy detailed in Figure 1. The new instrument was able to correct for any interferences, and it was FDA cleared for use in any care setting. This allowed clinical staff to obtain rapid results and enable immediate treatment. Hypoglycemic patients could be identified earlier, allowing quicker intervention and more effective care. This ease of appropriate use was very much appreciated compared to reviewing charts to verify the patient does not meet any of the exclusion criteria (hematocrit levels and medication review). It also reduced the number of samples sent to the main laboratory, which improved both patient and clinician satisfaction. Overall, faster treatment times improved patient care as well as throughput goals.
In the five years since transitioning to the new meter, there have been zero physician complaints regarding test results from the new glucose meter.
Operational and clinical benefits beyond expectations
We were surprised by the improved overall effectiveness of the new glucose meter. The previous meters had a number of electronic error codes that could only be fixed by a trained POC staff member or by replacing the instrument. With those codes, we saw approximately 10–20 instrument replacements monthly across all facilities. Each replacement was associated with time lost and additional efforts in quality control checks for incoming instruments. After implementation of the new interference-free glucometer, we observed a 50–75% reduction in replacements (3–5 per month). This was due to the end user having more access and ability to troubleshoot the instrument at the bedside without needing administrator intervention.
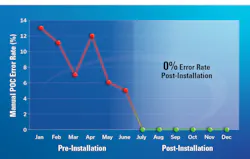
Another surprise benefit involved the manual test entry (MTE) tests. As mentioned above, our organization maintains an expansive POC program with a variety of waived tests on their menu. Some of the waived tests include urine pregnancy, whole blood pregnancy, rapid strep, visual urinalysis, gastric pH, and fecal occult blood. Recording these tests into the electronic medical record required either a form to be filled out or the results could be entered as an MTE into the glucometer, with the latter being more efficient, user friendly, and less error prone. With the previous glucometer only holding a maximum of five manual tests, the organization was restricted from easily capturing and transmitting data from all the necessary POC tests. Since there was no way for some of the tests to be uploaded to the electronic medical record without using an additional form, compliance issues were prominent. We noticed high monthly error rates for manual test entry of POC tests. For example, fingerstick pregnancy error rates were anywhere from 4–13% prior to implementation of the new instrument (Figure 2). Errors included unqualified staff performing tests, recording results, lack of lot number documentation, and improper expiration dates entered. After implementation, we were able to eliminate the external result form and the manual test entry error rate dropped to 0%, where it has remained (Figure 2). Another example was the gastric pH test in the neonatal ICU. The ability to capture 540/month previously undocumented results led to improved traceability and compliance.
Finally, there was a positive financial impact post transition. Fewer meter replacements reduced strip usage for linearity and quality control. Analyzing a six-month time frame pre- and post-installation, our overall glucose strip usage for quality controls was reduced by 17% in the Intensive Care Unit (ICU) and by 48% in the ICU step-down unit (or Intermediate Care). Similarly, test strips used for linearity were reduced by 33%.
How one technology upgrade improved the POC testing program
This case illustrates that investing in accurate, interference‑free technology pays dividends across multiple dimensions of patient care and operations. Overall, transitioning to the new glucose meter technology delivered measurable improvements across our multi‑hospital system, from streamlined workflows and reduced policy burden to higher staff satisfaction, lower costs, and increased compliance. By improving the performance of multiple POC testing workflows, the impact extended well beyond glucose testing.
References
- Klonoff DC, Vigersky RA, Nichols JH, Rice MJ. Timely hospital glucose measurement: Here today, gone tomorrow? Mayo Clin Proc. 2014;89(10):1331-5. doi:10.1016/j.mayocp.2014.08.005.
- Gaines AR, Pierce LR, Bernhardt PA. Fatal iatrogenic hypoglycemia: Falsely elevated blood glucose readings with a point-of-care meter due to a maltose-containing intravenous immune globulin product. U.S. Food and Drug Administration's Center for Biologics Evaluation and Research. Accessed March 11, 2026. https://web.archive.org/web/20080517085110/https://www.fda.gov/cber/safety/glucfalse.htm.
- Korsatko S, Ellmerer M, Schaupp L, et al. Hypoglycaemic coma due to falsely high point-of-care glucose measurements in an ICU-patient with peritoneal dialysis: A critical incidence report. Intensive Care Med. 2009;35(3):571-2. doi:10.1007/s00134-008-1362-7.
- Souza SP, Castro MCR, Rodrigues RA, Passos RH, Ianhez LE. False hyperglycemia induced by polivalent immunoglobulins. Transplantation. 2005;80(4):542-543. doi:10.1097/01.tp.0000168752.55185.74.
- Kirrane BM, Duthie EA, Nelson LS. Unrecognized hypoglycemia due to maltodextrin interference with bedside glucometry. J Med Toxicol. 2009;5(1):20-3. doi:10.1007/BF03160976.
About the Author

Cody Maddox, MS-CLS, MLS(ASCP)CM
is a Laboratory Manager at Grace Medical Center in Baltimore, Maryland. He oversees point-of-care testing for the LifeBridge Health system in Baltimore. Additionally, he serves as an adjunct professor for the Medical Laboratory Science program at Stevenson University.