Automated and comprehensive 25 VitDs measurement
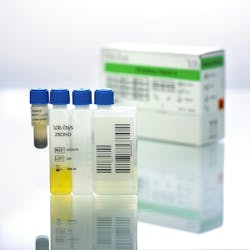
The IDS 25 VitDs assay (IS-2500N) quantitively measures total 25-hydroxyvitamin D [25(OH)D] in human serum and plasma on the IDS System, with co-specificity for 25(OH)D3 and 25(OH)D2. It is standardized to the ID-LCMS/MS Reference Procedure. Stable, ready-to-use human serum calibrators (IS-2520N) and controls (IS-2530N) are also available.
Vitamin D assay quality control
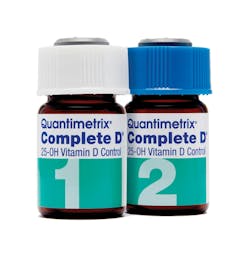
Complete D 25-OH Vitamin D Control is a liquid, ready-to-use quality control material for monitoring clinical assays measuring total 25-hydroxyvitamin D. Prepared in a human serum matrix with 25-OH vitamin D2 and D3, it represents clinically relevant concentrations and is suitable for daily quality control and comparison between test systems.
Comprehensive QC for vitamin D testing
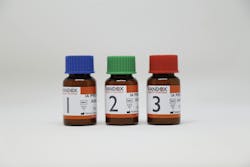
The Acusera Immunoassay Premium Plus control provides third‑party assessment for 54 analytes. Including 1-25‑(OH)₂ Vitamin D and 25‑OH Vitamin D, alongside routinely run tumor markers, hormones, and more in a lyophilized, human serum matrix.
Phosphorus-SL for vitamin D monitoring
SEKISUI’s Phosphorus assay is intended for the in vitro quantitative measurement of inorganic phosphorus in serum. Increased phosphorus concentrations (hyperphosphatemia) are usually the result of Vitamin D overdose, hypoparathyroidism, or renal failure. Decreased serum concentrations (hypophosphatemia) usually result from rickets, hyperparathyroidism, or Fanconi Syndrome.
CDC certified with defined pediatric expected values
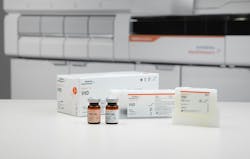
The Vitamin D Total Assay from Siemens Healthineers is the only CDC certified method with defined pediatric expected values. With defined pediatric reference ranges for the Atellica IM and ADVIA Centaur Vitamin D Total assays, ranges can be easily adopted with a simple in-lab validation*.
*Refer to CLSI EP28-A3c for recommendations on establishing and verifying reference ranges
Proficiency testing program for vitamin D
Clinical laboratories can ensure testing accuracy with WSLH PT's Vitamin D proficiency testing program. This program is included in the Immunoassay Chemistry B panel, provided in staggered shipments of lyophilized serum samples. For more details, refer to PT01712 on page 17 of the 2026 Clinical PT Catalog: wslhpt.org.