From informatics to AI: Building intelligent laboratory quality systems
To take the test online go HERE. For more information, visit the Continuing Education tab.
LEARNING OBJECTIVES
Clinical laboratories have always been quality-driven organizations, but the way quality is achieved is changing quickly. For much of the twentieth century, the center of gravity for quality management was the analytical phase: accuracy, precision, and reproducibility demonstrated through quality control materials, calibration verification, proficiency testing, and routine instrument maintenance. This approach established the laboratory’s reputation as a scientific environment where performance could be measured, controlled, and improved.
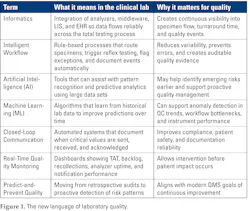
Over the past two decades, however, the definition of quality has expanded well beyond analytic performance. The modern quality management system (QMS) must encompass the entire total testing process, from ordering and specimen collection through accessioning, analysis, verification, reporting, and clinician response. In practice, many of today’s quality failures originate outside the analytical phase—in specimen labeling, transport delays, interface mapping errors, or communication breakdowns. Informatics has therefore become a core discipline of quality management, designing workflows that prevent errors, standardizing decisions, reducing variability, and capturing evidence automatically.
The next step in that evolution is the potential introduction of artificial intelligence (AI) and machine learning (ML) into laboratory quality management. While fully AI-driven quality systems are still emerging in most clinical laboratories, informatics-driven workflows are already transforming how laboratories monitor quality and manage risk.
AI is not a replacement for scientific judgment, nor is it a shortcut to quality. Its real value lies in its potential to make quality systems more proactive by helping detect patterns that humans may not easily see in real time. Used thoughtfully, AI can help laboratories transition from a reactive “inspect-and-correct” model to a proactive, data-driven “predict-and-prevent” approach.
From paper logs to smart data systems: The quiet revolution in laboratory quality
A useful way to understand the present moment is to look at how dramatically laboratory quality work has already changed. In the not-so-distant past, quality programs relied heavily on paper logs, manual temperature recordings, phone calls for critical values, handwritten corrective action notes, and retrospective audits to confirm compliance. The quality manager’s quality tools were often a collection of binders—QC records, maintenance logs, reagent inventories, competency checklists, and incident reports.
Today, middleware rules enforce autoverification thresholds, delta checks, specimen integrity flags, and instrument-specific quality hard stops. Digital traceability can capture time-stamped events from collection through reporting.
When a critical value is resulted, the preferred pathway is no longer a manual phone call with variable documentation; it is an integrated, closed-loop communication workflow that documents notification automatically. These systems reduce transcription risk, improve documentation reliability, and generate structured data that laboratories can analyze to identify improvement opportunities.
Informatics is the bridge between regulatory mandates and future quality systems, making the measurement of laboratory quality truly continuous. This shift—from paper to digital workflow—represents the real foundation on which future AI capabilities will build. For modern laboratory quality management terms, see Figure 1.
A practical example of informatics-enabled quality improvement: Epic Secure Messaging and critical value closed-loop communication
The benefits of informatics become clearest when viewed through a real quality problem. Critical value reporting has historically been one of the most labor-intensive and failure-prone post-analytic processes. Phone calls can be delayed by busy clinical units, unavailable clinicians, and inconsistent documentation. Implementing Epic Secure Messaging can help laboratories transition from manual communication to a closed-loop process that documents when a message is sent, opened, and acknowledged. This approach improves compliance, reduces transcription risk, and creates reliable data that can be analyzed for continuous quality improvement.
A laboratory’s implementation of Epic Secure Messaging illustrates what intelligent quality looks like in practice. Instead of relying on manual call chains, the system routes the result directly to the responsible provider, captures the exact time and user acknowledgment, and places the documentation automatically into the medical record. The workflow becomes standardized, auditable, and measurable transforming a historically inconsistent process into a controlled quality system.
Closed-loop communication reduces documentation gaps and produces structured data that laboratories can analyze by unit, service line, or time of day to identify trends and improvement opportunities. The benefits are not only faster notification, and better notification, but improved patient safety.
Real-world examples of emerging AI applications in laboratory quality
A common myth to be dispelled is that AI will replace laboratorians. In the clinical laboratory, AI can be a tool that supports—not replaces—professional judgement. It refers to tools that assist with pattern recognition, anomaly detection, and predictive analytics. Machine learning models can be trained on historical data to identify emerging trends in instrument performance, specimen integrity, or workflow bottlenecks. Informatics integrates systems so that data flows reliably between analyzers, middleware, LIS, and EHR environments. Intelligent workflows combine these capabilities into rule-based or analytics-supported processes that route specimens, flag exceptions, and document key quality events automatically.
Several legitimate AI-supported use cases are already emerging in laboratory medicine. Patient-based, real-time quality control approaches use statistical and machine learning methods to detect subtle analytical drift. Digital pathology tools use image recognition to support diagnostic consistency and workload triage. Hematology image-analysis systems assist with abnormal cell detection. Predictive maintenance analytics are being explored to identify patterns that precede analyzer downtime. Natural language processing tools can analyze incident reports to identify recurring quality risks.
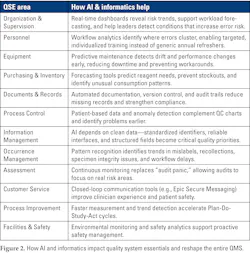
These applications are not yet universal, but they illustrate how AI may build upon existing informatics-driven quality systems. See Figure 2 for how AI impacts quality system essentials (QSE).
Real-time data as the new quality indicator
In the traditional model, quality indicators were often reviewed monthly or quarterly. That cadence can still be useful, but it is insufficient for an environment where specimen flow and analyzer demand can change hour by hour.
Modern laboratories increasingly depend on real-time visibility: turnaround time distributions, specimen queue backlogs, analyzer uptime, pending lists, incident trends, etc. This visibility changes quality culture. Instead of discovering problems in a retrospective audit, teams can intervene during the shift. The goal is to make corrections before patient impact occurs. When this is done well, it creates operational excellence. Staff see the system’s behavior, understand where bottlenecks arise, and actively intervene to prevent impact.
This mirrors how high-performing industries outside healthcare operate. Consumer technology organizations continuously measure engagement and performance to optimize systems. The laboratory equivalent is not “engagement”—it is patient safety, timeliness, and reliability. The goal is the same: use data as a feedback engine to optimize outcomes.
What must be in place before AI improves laboratory quality
The most common barrier to AI-enabled quality improvement is not the model itself. It is the laboratory’s readiness in informatics fundamentals. If LIS-to-EHR interoperability is inconsistent, if specimen identifiers are not unified across systems, or if workflow steps are not captured as discrete events, AI has little stable ground to stand on. Laboratories must first modernize data capture and interface integrity before AI can deliver meaningful quality gains.
Another common barrier is metric overload. The QMS must prioritize indicators that are actionable and tied to improvement responsibilities. ISO-aligned thinking helps here: quality indicators should systematically monitor and evaluate contributions to patient care, not become an endless list of numbers.
Finally, culture matters. Building a data-driven culture is not about surveillance, it is about transparency and creating a learning environment. Quality improves when staff can see how the system works, trust the fairness of measurement, and are trained to interpret AI/informatics tools alongside their professional judgment. When teams understand how the tools work—and how to question them appropriately—they solve problems more effectively and make decisions with greater confidence.
Conclusion: The future of laboratory quality is intelligent
Clinical laboratory quality has always been about discipline: controlling processes, documenting performance, and improving continuously. What changes in the AI era is the laboratory’s ability to sense its own operation in real time, detect risk earlier, and optimize faster.
Informatics and intelligent workflows are already transforming quality management by automating data capture, standardizing decisions, and creating closed-loop evidence of compliance. AI will build upon that transformation by adding predictive surveillance, pattern detection across the total testing process, and operational optimization that reduces variability and error risk.
The laboratory of the future is not simply automated—it is intelligent, continuously learning from its own data and monitoring its systems, where technology strengthens rather than replaces professional judgment. In that environment, quality management becomes what it has always aspired to be: proactive, adaptive, continuous, and built into the workflow by design.
References
- Gilligan J. Transforming laboratory medicine through AI: From promise to practice. Mayo Clinic Laboratories. October 6, 2025. Accessed April 1, 2026. https://news.mayocliniclabs.com/2025/10/06/transforming-laboratory-medicine-through-ai-from-promise-to-practice/.
- Saul H. How AI and machine learning are revolutionizing the laboratory. Roche Diagnostics LabLeaders. October 1, 2025. Accessed April 1, 2026. https://diagnostics.roche.com/global/en/lab-leaders/article/machine-learning-ai-in-laboratory.html?.
- 42 CFR part 493 subpart K -- quality system for nonwaived testing. eCFR. Accessed April 1, 2026. https://www.ecfr.gov/current/title-42/chapter-IV/subchapter-G/part-493/subpart-K.
- Lorde N, Mahapatra S, Kalaria T. Machine learning for patient-based real-time quality control (PBRTQC), Analytical and preanalytical error detection in clinical laboratory. Diagnostics (Basel). 2024;14(16):1808. doi:10.3390/diagnostics14161808.
- Artificial intelligence risk management framework (AI RMF 1.0). National Institute of Standards and Technology. Accessed April 1, 2026. https://nvlpubs.nist.gov/nistpubs/ai/NIST.AI.100-1.pdf.
- Artificial intelligence/machine learning (AI/ML)-based software as a medical device (SaMD) action plan. Food and Drug Administration. January 2021. Accessed April 1, 2026. https://www.fda.gov/media/145022/download.
- Tan B. How to validate AI algorithms in anatomic pathology. College of American Pathologists. Accessed April 1, 2026. https://www.cap.org/member-resources/clinical-informatics-resources/how-to-validate-ai-algorithms-in-anatomic-pathology.
To take the test online go HERE. For more information, visit the Continuing Education tab.
About the Author

Nancy Ross MS, MT(ASCP)cmp, CQIA (ASQ), CLC (AMT), CQM/OE (ASQ)
is a U.S. Navy–trained clinical laboratory scientist with over 20 years of experience in hospital, reference, and academic laboratory settings. She is the founder of Improov Consulting, Inc., a laboratory consulting company focusing on performance improvement and quality.

Irwin Z. Rothenberg MBA, MS CLS (ASCP)
has over 40 years of experience in laboratory medicine. He previously worked as the technical writer / quality advisor at COLA Resources, Inc. and is now a freelance technical writer for Improov. Irwin has contributed articles to several professional publications including Physician Office Resources; AAFP-PT POL Insight; Medical Laboratory Observer; and COLA Insights.

Graham K. Mafela, MLS (ASCP), MBA
has over 20 years of experience in laboratory medicine and has been providing consulting services since 2012. He is ASCP board-registered as a Medical Laboratory Scientist and holds an MBA from Indiana Wesleyan University. He is the Chief Operations Officer for Improov Consulting.