Outpatient diagnostics, antimicrobial stewardship, and health equity: Closing gaps in the fight against antimicrobial resistance
To take the test online go HERE. For more information, visit the Continuing Education tab.
LEARNING OBJECTIVES
The spread of antimicrobial-resistant (AMR) infections poses a global public health threat, with over 1 million resistant infections diagnosed annually.1 With more than 200 million antimicrobials prescribed in outpatient settings, including primary care clinics, urgent care centers, emergency departments (ED), and skilled nursing facilities, it is evident that these treatment settings facilitate the spread of AMR infections.2,3 These are areas of unmet need as most antimicrobial stewardship (AMS) programs, designed to optimize unnecessary antimicrobial prescribing, have traditionally focused on addressing gaps in inpatient settings.4 Given the lack of oversight in outpatient AMS, it is unsurprising that there have been growing reports of inequities in the diagnosis and management of AMR infections, specifically among marginalized populations.3,5,6
While identifying as part of a marginalized group is not a biological risk factor for developing an AMR infection, marginalization is often accompanied by systems of oppression such as racism and classism.7 These systems can lead to inequities in the social determinants of health (SDoH), such as education, socioeconomic status (SES), and access to healthcare.8,9 For example, individuals who identify as racially and ethnically marginalized (REM) are underrepresented among college graduates.10 This highlights potential barriers to health literacy and understanding of diagnosis severity and instructions for antimicrobial use.11 Additionally, areas of low SES are more vulnerable to shortages in healthcare providers and associated resources.12 Ultimately, limited access to care can lead to treatment delays or the absence of treatment for chronic and acute infectious illnesses.
Integrating diagnostics and appropriate stewardship principles has revolutionized inpatient AMS programs.4 With the evolution of existing diagnostics and the emergence of innovative rapid diagnostics tests (RDTs), including rapid antigen tests (RADTs) and molecular diagnostics, there is an opportunity to optimize AMS in outpatient settings. Furthermore, point-of-care tests (POCTs), often defined as those RDTs not performed in a centralized laboratory and run with a Clinical Laboratory Improvement Amendments (CLIA) waiver or that are moderately complex, also have accelerated management capabilities the closer they get to the patient.13 Ultimately, if integrated and utilized appropriately, RDTs may lead to a reduction in antimicrobial usage and further inequitable impacts of resistant infections across marginalized groups. Here, we provide an overview of the Centers for Disease Control and Prevention (CDC) guidance on outpatient AMS programs and insight into the microbiology laboratory's role in integrating RDTs into AMS procedures. We also provide insight into the use of RDTs in outpatient settings. Finally, we conclude with strategies for integrating these diagnostic modalities into outpatient AMS programs to narrow AMR-related health equity gaps by reducing inappropriate antimicrobial prescribing and use.
Overview of the CDC outpatient antimicrobial stewardship framework
To address the estimated 30–50% of unnecessary prescriptions in the outpatient community, the CDC published ambulatory AMS guidance in 2016 with the following core elements: commitment, action for policy and practice, tracking and reporting, and education and expertise.14 Despite The Joint Commission endorsing outpatient stewardship initiatives the following year, widespread adoption has been slow due to barriers such as variable governance and data structures, diverse models of care, lack of administrative commitment, and disparities in resource allocations.15 A survey of Vizient member hospitals, representing 20% of the country’s ambulatory market, reported that only 7% had a fully functioning ambulatory AMS program compared to 88% of inpatient settings.16 Analogous to inpatient clinical laboratories, RDTs (including RADTs and molecular diagnostic tests) can augment AMS efforts in the outpatient setting and provide clinicians with timely and actionable information by differentiating commonly encountered pathogens implicated in the greatest driver of unnecessary outpatient antibiotic prescriptions: upper respiratory tract infections (URIs).
Diagnostic advancements
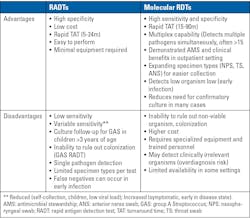
Rapid antigen diagnostic tests (RADTs)
RADTs are commonly used in point-of-care settings as they provide results within 5-30 minutes for frequent URI pathogens such as group A Streptococcus (GAS), influenza viruses, and SARS-CoV-2. These tests are inexpensive lateral flow immunoassays without specific storage constraints that produce a visible signal from migrating antigen-antibody complexes by capillarity.17 More information on RADTs is available in Table 1.
Many guidelines recommend using RADTs for GAS, which demonstrate pooled sensitivities and specificities of 85.6% and 95.4%, respectively, compared to throat culture.18 While high specificity may support actionable results when positive, the moderate sensitivity raises questions about back-up throat cultures in children aged three and older, negating the quick turnaround time, or whether the cause of pharyngitis is secondary to a viral pathogen or non-infectious etiology.
Similar to RDTs for bacterial pathogens, viral RADTs are also not without limitations. SARS-CoV-2 demonstrates pooled sensitivities and specificities of 71–82% and 99%, respectively; however, performance is dependent on several factors.19 Rapid influenza diagnostic tests (RIDTs) also report variable performance with sensitivities and specificities of 61–69% and 98–99%, respectively; however, real-world practice often records sensitivities as low as 25–50%.20 These poor sensitivities reduce diagnostic certainty, thereby limiting antibiotic prescriptive confidence resulting in antimicrobials “just in case” for negative tests and/or additional confirmatory PCR testing when clinical suspicion is high.
Molecular diagnostic tests
To overcome some limitations of RADTs, molecular RDTs for GAS and respiratory viruses use either thermocycling or isothermal amplification, with PCR being the most common thermocycling method. Newer RDTs employ isothermal techniques such as Nicking Enzyme Amplification Reaction, Loop-Mediated Isothermal Amplification, and Helicase-Dependent Amplification.18 Pooled sensitivities and specificities are significantly higher at 97.5% and 95.1%, respectively, for GAS.21,22 Sensitivities for viral analytes may be as high as 100% depending on the analyte and test.22 Certain molecular RDTs can simultaneously detect viral and bacterial targets from nasopharyngeal, anterior nares, and throat swabs, providing integrated sample-to-answer systems that identify multiple pathogens within 15–90 minutes.23
Molecular tests in point-of-care settings
Recent data suggest promising outcomes, particularly among marginalized patients, when newer POCT diagnostics are leveraged in the outpatient space. Meltzer and colleagues compared a multiplex respiratory POCT with a ~15-minute turnaround time to an observational control for the management of URIs in the ED.24 Notably, REM patients were less likely to receive antibiotics with confirmed viral infections (6.5% versus 20.2%; p=0.009) and median length of ED stay was shortened (4.3 hours versus 6.5hours; p<0.001). Moreover, providers noted increased diagnostic confidence, and patients reported higher satisfaction with result timeliness. Utilizing the same POCT, improved patient outcomes and reduced prescriptions were reproducible in additional outpatient cohorts.25,26 Based on these recent studies, molecular RDTs offer significant opportunities for reducing unnecessary antibiotic use in the outpatient arena to combat AMR; however, notable barriers to increase community access and AMS integration need to be considered.
Current and anticipated gaps in the use of RDTs in outpatient settings
The COVID-19 pandemic exposed barriers that can hinder the equitable use of RDTs to enhance outpatient AMS initiatives. By using deprivation indices like the social vulnerability index (SVI) and area deprivation index (ADI), which assess the effects of social, material, and economic disadvantages on health outcomes, researchers have obtained intriguing findings.27-29 Several studies have shown disparities in the availability of RDTs, with high social vulnerability areas less likely to have access to test-to-treat locations; a situation likely to worsen due to shifting government funding priorities.30,31
The shortage of test-to-treat sites is a significant concern because socially vulnerable areas—often inhabited by marginalized groups—are expected to face increased burdens from vaccine-preventable illnesses in upcoming viral seasons. This is due to changes in the dissemination of reliable vaccine information and evolving vaccine recommendations, which could influence the future accessibility and affordability of immunizations. These health systems, frequently categorized as safety-net institutions, tend to be understaffed and underfunded.32 Thus, an increase in the number of individuals needing testing could further strain an already fragile system. Consequently, this overburden could increase the likelihood of diagnostic errors among vulnerable populations.33 Such errors could lead to delayed or missed treatment of viral infections, potentially fostering the development of infections caused by AMR bacteria.33 Even more concerning, these limitations in diagnosis and management might erode trust in future clinician diagnoses and the perceived effectiveness of antimicrobial therapy prescribed in outpatient settings.33
Other operational concerns regarding the deployment of RDTs in outpatient settings were also raised during the pandemic.34 These concerns include difficulty in complying with the manufacturers’ specifications and storage requirements, staff training and competency validation, and regulatory compliance with laboratory accreditation and regulatory standards. These issues are likely to be more prevalent and harder to address in outpatient settings in vulnerable areas, where healthcare shortages and turnover are most pronounced.
Financial reimbursement for RDTs is also a point of contention, as batch testing is often cheaper.34 There may be variability in the tests for which insurance programs are willing to provide coverage, which may affect the reliability of the results and the treatment prescribed based on the test output.
Strategies, solutions, and how to augment current successes in outpatient antimicrobial stewardship efforts
Integrating RDTs into outpatient AMS programs can play a major role in addressing inequities in AMR infections. Nonetheless, this effort will not be without challenges. Therefore, in this section we provide evidence-based strategies for clinicians to consider as guidance for the equitable inclusion of RDTs in outpatient stewardship programs.
Notable resource-limited interventions include antibiotic commitment posters or screensavers, delayed prescribing prescription pads, and signed antibiotic commitment letters by office and clinic staff.35,36 With increased sensitivity of molecular RDTs compared to RADTs, one increasingly utilized strategy is pairing clinical prediction rules with patient assessment. One group embedded the Centor score into the electronic health record with pharyngitis testing to improve pre-test probability.37
While information technology plays a key role in AMS success, not every outpatient setting may have this resource. Risk assessment scoring tools could be available in posted guidance documents for pharyngitis, which would align with the recently updated Infectious Diseases Society of America (IDSA) GAS Pharyngitis guideline.38 It is outside the scope of this article to list all supplemental strategies for outpatient stewardship programs, though additional resources can be found on the CDC’s Outpatient Stewardship and the Agency for Healthcare Research and Quality (AHRQ) websites.
Another notable barrier to successful instrumentation deployment is lack of awareness of availability of testing and the valuable information and outcomes that can be harnessed from specific platforms for key stakeholders (e.g., providers, patients and care givers, insurers). Outpatient providers believe published local resistance patterns would incentivize ambulatory stewardship implementation.39 Local epidemiology could come from state health departments, regional clinical microbiology laboratories, or de-identified reports directly from RDTs platforms. One such surveillance system is BioFire Syndromic Trends by bioMérieux who publishes respiratory pathogen and gastrointestinal trends publicly. This data provides near real-time information on circulating pathogens to guide management decisions.40 Compared to the CDC’s Respiratory Virus Hospitalization Surveillance Network (RESP-NET) detailing national trends for influenza, COVID-19, and RSV, Syndromic Trends includes results with additional viruses (e.g., adenovirus, seasonal coronaviruses, entero/rhino virus, parainfluenza, human metapneumovirus) from a comprehensive multiplex panel directly from U.S. clinical laboratories using this technology.
Patients and caregivers are also major determinants of antibiotic prescriptions in the community. While this is multifactorial, one potential driver is that the public is largely unaware of the risks associated with antibiotic use, especially those with marginalized identities.41 Nearly 4 in 5 providers agree that stewardship efforts would be ineffective unless paired with efforts to educate patients on resistance and the importance of de-prescribing antibiotics for viruses.39
In addition to clinic stewardship commitment pledges, providers can explain the value of broader multiplex panels that help detect yearly circulating respiratory viruses like rhinovirus and enterovirus. By providing targets beyond the traditional quadraplex (e.g., Influenza A/B, SARS-CoV-2, RSV), 44% more detections were identified in one study with a multiplex system, which allows for more comprehensive management and de-prescribing of antibiotics for viral infections.42 Lastly, external market access stakeholders (e.g., government and regulatory agencies, payors) must be informed of the clinical value and patient benefit of IVD technology in the outpatient setting for updating coverage policies associated with test utilization.43
For addressing RDT accessibility concerns, the Prioritizing Equity in Antimicrobial Stewardship Efforts (EASE) framework, aligned with Joint Commission and CMS Health Equity principles may represent an effective strategy.44 The framework, which includes five focus areas, states that reviewing institutional data should be the starting point when designing an intervention to create change. Following this guidance, investigators could identify an outpatient setting and a disease area to review data for. With the growing reports of S. pneumoniae inequities in the diagnosis of AMR S. pneumoniae infections, this may be an ideal starting point for clinicians and researchers.6 Following the guidance provided in the EASE framework, researchers could begin with extracting data on S. pneumoniae infections treated in their affiliated outpatient settings. The investigators could further disaggregate the data by levels of deprivation (using the ADI or SVI), race/ethnicity, or insurance status to see whether differences in AMR S. pneumoniae infections exist. Investigators have used non-susceptibility to at least one of the 27 antimicrobials clinically used to treat pneumococcal infections as specified in the National Healthcare Safety Network (NHSN) Antimicrobial Use and Resistance (AUR) reporting module as an indicator for S. pneumoniae resistance, which other researchers can consider as their definition for an AMR S. pneumoniae infection.45 Based on these findings, investigators and clinicians can design an intervention to address uncovered inequities, with accessibility as a focus. The intervention should consider operational barriers, including staff workload and competency. Costs and insurance coverage should also be evaluated when deciding who should be tested, when tests should be used, and which RDTs should be prioritized for use. With these considerations, a possible intervention could involve deploying RDTs only for individuals at higher risk for AMR pneumococcal infections. This approach could ensure that patients at increased risk receive the right antimicrobials promptly, thereby reducing the development and propagation of resistant infections among high-risk groups.
Conclusion
The evolution of diagnostics for infectious diseases has increased accuracy, reduced turnaround times, and broadened the spectra of detections with one test. RDTs boost diagnostic certainty, translating into improved antimicrobial use and clinical outcomes in the inpatient and outpatient settings alike. Health disparities present predictable barriers to ensuring access to RDTs in marginalized populations, though these challenges can be met. The spread of AMR is not constrained by geography or socioeconomic status. Availability of outpatient RDTs for all communities ultimately align with the CDC’s Ambulatory Stewardship guidance, strengthening antibiotic prescribing practices and protecting population health at large—reflecting the principle: “Whatever affects one directly, affects all indirectly.”
REFERENCES
1. GBD 2021 Antimicrobial Resistance Collaborators. Global burden of bacterial antimicrobial resistance 1990-2021: A systematic analysis with forecasts to 2050. Lancet. 2024;404(10459):1199-1226. doi:10.1016/S0140-6736(24)01867-1.
2. Outpatient Antibiotic Prescriptions - United States, 2022. CDC. November 13, 2023. Accessed January 22, 2026. https://archive.cdc.gov/#/details?url=https://www.cdc.gov/antibiotic-use/data/report-2022.html.
3. Thure KA, Mutamba G, Wren CM, Evans CD. Assessing disparities in inappropriate outpatient antibiotic prescriptions in Tennessee. Antibiotics (Basel). 2025;14(6):569. doi:10.3390/antibiotics14060569.
4. Claeys KC, Johnson MD. Leveraging diagnostic stewardship within antimicrobial stewardship programmes. Drugs Context. 2023;12:2022-9-5. doi:10.7573/dic.2022-9-5.
5. McLeod CC, Al-Fayiz H, Rodriguez S, Tan KK, Abdul-Mutakabbir JC. Examining racial and social vulnerability disparities in the outpatient treatment of uncomplicated cystitis at a Southern California Academic Hospital. Antimicrob Steward Healthc Epidemiol. 2023;3(1):e214. doi:10.1017/ash.2023.469.
6. Mohanty S, Ye G, Sheets C, et al. Association between social vulnerability and streptococcus pneumoniae antimicrobial resistance in US adults. Clin Infect Dis. 2024;79(2):305-311. doi:10.1093/cid/ciae138.
7. Abdul-Mutakabbir JC, Abdul-Mutakabbir R. Syndemics of antimicrobial resistance: Non-communicable diseases, social deprivation, and the rise of multidrug-resistant infections. Infect Dis Ther. 2025;14(8):1561-1575. doi:10.1007/s40121-025-01188-1.
8. Brown TH, Homan P. The future of social determinants of health: Looking upstream to structural drivers. Milbank Q. 2023;101(S1):36-60. doi:10.1111/1468-0009.12641.
9. Marcelin JR, Hicks LA, Evans CD, et al. Advancing health equity through action in antimicrobial stewardship and healthcare epidemiology. Infect Control Hosp Epidemiol. 2024;45(4):412-419. doi:10.1017/ice.2024.7.
10. Libassi CJ. The neglected college race Gap: Racial disparities among college completers. CAP. May 23, 2018. Accessed January 22, 2026. https://www.americanprogress.org/article/neglected-college-race-gap-racial-disparities-among-college-completers/.
11. Muvuka B, Combs RM, Ayangeakaa SD, et al. Health literacy in African-American communities: Barriers and strategies. Health Lit Res Pract. 2020;4(3):e138-e143. doi:10.3928/24748307-20200617-01.
12. Jones CH, Dolsten M. Healthcare on the brink: Navigating the challenges of an aging society in the United States. NPJ Aging. 2024;10(1):22. doi:10.1038/s41514-024-00148-2.
13. Lisby JG, Schneider UV. Point of care testing for infectious disease: Ownership and quality. J Antimicrob Chemother. 2021;76(Suppl 3):iii28-iii32. doi:10.1093/jac/dkab247.
14. Sanchez GV, Fleming-Dutra KE, Roberts RM, Hicks LA. Core elements of outpatient antibiotic stewardship. MMWR Recomm Rep. 2016;65(6):1-12. doi:10.15585/mmwr.rr6506a1.
15. Frost HM, Hersh AL, Hyun DY. Next steps in ambulatory stewardship. Infect Dis Clin North Am. 2023;37(4):749-767. doi:10.1016/j.idc.2023.07.004.
16. Eudy JL, Pallotta AM, Neuner EA, et al. Antimicrobial stewardship practice in the ambulatory setting from a national cohort. Open Forum Infect Dis. 2020;7(11):ofaa513. doi:10.1093/ofid/ofaa513.
17. Budd J MB, Weckman NE, et al. Lateral flow test engineering and lessons learned from COVID-19. Nat Rev Bioeng. 2023;1:13-31. doi:https://doi.org/10.1038/s44222-022-00007-3.
18. Cohen JF, Tanz RR, Shulman ST. Group A streptococcus pharyngitis in children: New perspectives on rapid diagnostic testing and antimicrobial stewardship. J Pediatric Infect Dis Soc. 2024;13(4):250-256. doi:10.1093/jpids/piae022.
19. Bachelet VC, Lizana FJ, Andrades CO, et al. Estimates for diagnostic accuracy of rapid antigen tests for SARS-CoV-2 in systematic reviews are consistently similar despite poor methodological rigor: a methodological overview. J Clin Epidemiol. 2024;176:111547. doi:10.1016/j.jclinepi.2024.111547.
20. Nairz M, Weiss G. How to identify respiratory pathogens in primary health care - a review on the benefits, prospects and pitfalls in using point of care tests. Infection. 2025;53(6):2321-2340. doi:10.1007/s15010-025-02600-1.
21. Dubois C, Smeesters PR, Refes Y, et al. Diagnostic accuracy of rapid nucleic acid tests for group A streptococcal pharyngitis: systematic review and meta-analysis. Clin Microbiol Infect. 2021;27(12):1736-1745. doi: 10.1016/j.cmi.2021.04.021.
22. Matic N, Lawson T, Ritchie G, Lowe CF, Romney MG. Testing the limits of multiplex respiratory virus assays for SARS-CoV-2 at high cycle threshold values: Comparative performance of cobas 6800/8800 SARS-CoV-2 & Influenza A/B, Xpert Xpress SARS-CoV-2/Flu/RSV, and cobas Liat SARS-CoV-2 & Influenza A/B. J Assoc Med Microbiol Infect Dis Can. 2024;8(4):328-335. doi:10.3138/jammi-2022-0039.
23. Information on rapid molecular assays, RT-PCR, and other molecular assays for diagnosis of influenza virus infection. CDC. Oct 21, 2019. Accessed January 23, 2026. https://www.cdc.gov/flu/hcp/testing-methods/molecular-assays.html.
24. Meltzer AC, Payette C, Heidish R, et al. Point-of-care respiratory diagnosis and antibiotic utilization in the emergency department: A prospective evaluation of multiplex PCR. Acad Emerg Med. 2026;33(1):e70156. doi: 10.1111/acem.70156.
25. Bigaud B, Marjanovic N, Deroche L, et al. Impact of multiplex PCR point-of-care platform implementation for respiratory pathogen detection in an emergency department with high daily patient volume. J Clin Microbiol. 2026;64(1):e0131325. doi:10.1128/jcm.01313-25.
26. Arnold CG, Furtado T, Bang H, Harnett G, May LS. Combining an antibiotic stewardship program with a 15-pathogen viral panel to reduce inappropriate antibiotic prescribing. Microbiol Spectr. 2026;14(1):e0219525. doi:10.1128/spectrum.02195-25.
27. Boehmer TK, Koumans EH, Skillen EL, et al. Racial and Ethnic Disparities in Outpatient Treatment of COVID-19 - United States, January-July 2022. MMWR Morb Mortal Wkly Rep. 2022;71(43):1359-1365. doi:10.15585/mmwr.mm7143a2.
28. Mullachery PH, Li R, Melly S, et al. Inequities in spatial accessibility to COVID-19 testing in 30 large US cities. Soc Sci Med. 2022;310:115307. doi:10.1016/j.socscimed.2022.115307.
29. Meyer AND, Giardina TD, Khawaja L, Singh H. Patient and clinician experiences of uncertainty in the diagnostic process: Current understanding and future directions. Patient Educ Couns. 2021;104(11):2606-2615. doi:10.1016/j.pec.2021.07.028.
30. Hohenstein L, Maloney M, Banach DB. Paxlovid utilization and social vulnerability: trends in Connecticut from 2022 to 2023. Antimicrob Steward Healthc Epidemiol. 2025;5(1):e275. doi:10.1017/ash.2025.10193.
31. Smith ER, Oakley EM. Geospatial Disparities in Federal COVID-19 Test-to-Treat Program. Am J Prev Med. 2023;64(5):761-764. doi:10.1016/j.amepre.2023.01.022.
32. McHugh MD, Brooks Carthon M, Sloane DM, et al. Impact of nurse staffing mandates on safety-net hospitals: Lessons from California. Milbank Q. 2012;90(1):160-86. doi:10.1111/j.1468-0009.2011.00658.x.
33. McDonald KM, Gleason KT, Grob RN, et al. Exploring sociodemographic disparities in diagnostic problems and mistakes in the quest for diagnostic equity: Insights from a national survey of patient experiences. Front Public Health. 2025;13:1444005. doi:10.3389/fpubh.2025.1444005.
34. Oyefolu O, Gronvall GK. Exploring challenges and policy considerations in point-of-care testing for hospital preparedness ahead of infectious disease emergencies: A qualitative study. Infect Dis Health. 2025;30(2):111-118. doi:10.1016/j.idh.2024.10.001.
35. Meeker D, Knight TK, Friedberg MW, et al. Nudging guideline-concordant antibiotic prescribing: a randomized clinical trial. JAMA Intern Med. 2014;174(3):425-31. doi:10.1001/jamainternmed.2013.14191.
36. Core elements of outpatient antibiotic stewardship. CDC. August 21, 2025. Accessed January 23, 2026. https://www.cdc.gov/antibiotic-use/hcp/core-elements/outpatient-antibiotic-stewardship.html.
37. Aalbers J, O'Brien KK, Chan WS, et al. Predicting streptococcal pharyngitis in adults in primary care: a systematic review of the diagnostic accuracy of symptoms and signs and validation of the Centor score. BMC Med. 2011;9:67. doi:10.1186/1741-7015-9-67.
38. Linder JA, Watson ME, Wessels MR, et al. 2025 Clinical Practice Guideline Update by the Infectious Diseases Society of America on Group A Streptococcal (GAS) Pharyngitis: Risk assessment using clinical scoring systems in children and adults. Clin Infect Dis. 2025:ciaf668. doi:10.1093/cid/ciaf668.
39. Zetts RM, Garcia AM, Doctor JN, et al. Primary care physicians' attitudes and perceptions towards antibiotic resistance and antibiotic stewardship: A national survey. Open Forum Infect Dis. 2020;7(7):ofaa244. doi:10.1093/ofid/ofaa244.
40. Meyers L, Ginocchio CC, Faucett AN, et al. Automated real-time collection of pathogen-specific diagnostic data: Syndromic infectious disease epidemiology. JMIR Public Health Surveill. 2018;4(3):e59. doi:10.2196/publichealth.9876.
41. Szymczak JE, Klieger SB, Miller M, Fiks AG, Gerber JS. What parents think about the risks and benefits of antibiotics for their child's acute respiratory tract infection. J Pediatric Infect Dis Soc. 2018;7(4):303-309. doi:10.1093/jpids/pix073.
42. Banerjee D, Sasidharan A, Gummersheimer S, et al. Head-to-head comparison of the performance of BIOFIRE(R) SPOTFIRE(R) Respiratory/Sore Throat Panel, Cepheid Xpert(R) Xpress SARS-CoV-2/Flu/RSV and Cobas(R) SARS-CoV-2 & Influenza A/B assay for detection of respiratory viruses. J Clin Virol. 2025;182:105902. doi:10.1016/j.jcv.2025.105902.
43. Hill BK, Prinzi AM, Kane JR, Corby AK, Powe BD. Navigating and advancing market access for in vitro diagnostics: Understanding the roles of key stakeholders and policy. Open Forum Infect Dis. 2025;12(Suppl 2):S1404-S1412. doi:10.1093/ofid/ofaf444.
44. Abdul-Mutakabbir JC, Tan KK, Johnson CL, et al. Prioritizing equity in antimicrobial stewardship efforts (EASE): A framework for infectious diseases clinicians. Antimicrob Steward Healthc Epidemiol. 2024;4(1):e74. doi:10.1017/ash.2024.69.
45. Rozenbaum MH, Tort MJ, Chapman R, et al. Conceptual methodological framework for incorporating antimicrobial resistance considerations in economic models for pneumococcal conjugate vaccines. Infect Dis Ther. 2025;14(12):2853-2868. doi:10.1007/s40121-025-01243-x.
To take the test online go HERE. For more information, visit the Continuing Education tab.
About the Author

Abdullah Kilic, MD, D(ABMM)
is an Assistant Professor in Clinical Microbiology at Wake Forest School of Medicine. He is board-certified by the American Board of Medical Microbiology and has extensive experience in antimicrobial resistance, rapid diagnostics, and molecular microbiology. His research focuses on improving diagnostic accuracy and antimicrobial stewardship through advanced laboratory methods.

Jacinda (JAM) Abdul-Mutakabbir PharmD, MPH
is an Associate Professor of Clinical Pharmacy at UC San Diego whose research focuses on antimicrobial resistance, stewardship, and health disparities in infectious diseases. She has published more than 70 peer-reviewed articles and has received numerous national and international honors, including awards from IDSA, ACCP, SIDP, ESCMID, and the U.S. Public Health Service. She currently serves as an editor for Clinical Microbiology Reviews and is an active member of ACCP, ASM, IDSA, and SHEA.

Dusten T. Rose, PharmD, BCIDP
is a Medical Science Liaison in U.S. Medical Affairs at bioMérieux. He is a board-certified infectious diseases pharmacist with nearly 15 years of inpatient clinical experience. His interests include antimicrobial stewardship, clinical impact of emerging diagnostics, and antimicrobial resistance.