Diagnostic considerations with inherited platelet disorder testing
CONTINUING EDUCATION
Please read Article I and Article II before taking the test. To earn CEUs, visit www.mlo-online.com under the CE Tests tab.LEARNING OBJECTIVES
1. Identify complications with platelet disorders and populations at risk.
2. Define and identify the genetic pathology of platelet function disorders.
3. Define and identify the genetic pathology of inherited platelet disorders.
4. Discuss thalassemia pathology and the importance of genetic testing.
Inherited platelet disorders (IPDs) are a heterogeneous group of conditions that can affect platelet number, platelet function, or both. Symptoms may include purpura, petechiae, prolonged bleeding from cuts, epistaxis, gum bleeding, excessive bleeding after surgery, hemoptysis, hematuria, and menorrhagia in women. Severe platelet disorders and those with clear syndromic associations can be evident in the newborn period and early childhood, while mild thrombocytopenia may remain undiagnosed until adulthood when it is detected incidentally on routine blood counts.
Misdiagnosis of platelet disorders can result in inappropriate therapies and inadequate surveillance for additional medical complications. The diagnosis of a suspected IPD may be difficult to establish based solely on functional studies, especially in patients with milder disorders, as these assays are often technically challenging and difficult to interpret, and typically require immediate testing on fresh patient platelets due to limited sample stability. Advances in genetic testing through next-generation sequencing (NGS) allow for identification of underlying genetic defects and for distinguishing inherited platelet disorder cases from acquired platelet disorders, including immune thrombocytopenia. Accurate diagnosis is essential in guiding medical management of bleeding and associated non-hematologic conditions, assisting with the identification of affected family members, and predicting genetic recurrence risk assessment.
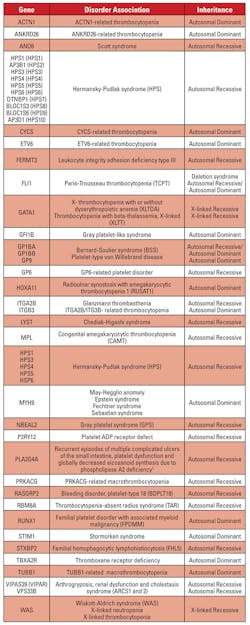
While this review will focus primarily on hallmark platelet disorders, a summary table on gene-associated clinical phenotypes is also provided (Table 1).
PLATELET FUNCTION DISORDERS
Inherited platelet function disorders are typically associated with normal platelet counts and may be variable in severity. They are caused by defects in platelet adhesion, glycoprotein expression, receptor function, signaling pathways, aggregation, cytoskeleton proteins, secretion, or granular contents and abnormalities in procoagulant activity. It should be noted that abnormal platelet function may be acquired by use of medication or secondary to certain medical conditions; these should be accounted for in the differential diagnosis.
Disorders of platelet adhesion
Bernard-Soulier Syndrome (BSS) is a rare inherited bleeding disorder due to absence or dysfunction of the platelet glycoprotein receptor Ib/V/IX complex. Laboratory evaluation typically reveals mild to moderate thrombocytopenia, unusually large platelets, and abnormal platelet function with absent or markedly reduced aggregation response to ristocetin. Flow cytometric analysis demonstrates absence of GPIb (CD 42) on the platelet surface. GPIbα, GPIbβ and GPIX, encoded by the GP1BA, GP1BB and GP9 genes, are all required for efficient expression of the complex on the platelet surface, while absence of GPV does not appear to affect receptor expression or VWF binding.2 Most cases of BSS are inherited as an autosomal recessive genetic trait, but autosomal dominant variants have been reported.
Disorders of platelet aggregation
Glanzmann thrombasthenia (GT) is caused by an abnormality in the genes which encode glycoproteins IIb/IIIa, the glycoprotein IIb/IIIa receptor (also called the fibrinogen receptor). Absence or dysfunction of the fibrinogen receptor does not allow for normal coagulation and presents as a bleeding phenotype. Laboratory evaluation typically reveals normal platelet counts and morphology. Platelet aggregation is highly specific for GT, as platelet aggregation fails to occur with any agonist, except ristocetin, where the reaction is preserved.3 Flow cytometric analysis generally demonstrates decreased quantity or absence of GPIIb/IIIa on the platelet surface. Genetic analysis has shown autosomal recessive pathogenic variants in the ITGA2B or ITGB3.
Disorders of platelet secretion
Platelet activation involves the release of several cytoplasmic granules. When released, alpha granules, dense granules, and lysosomes secrete their contents to assist in the adhesion of segregation of platelets.
Gray platelet syndrome (GPS) is an alpha granule deficiency typically characterized by large platelets, mild to moderate thrombocytopenia, and bleeding caused by homozygous or compound heterozygous mutations in the NBEAL2.4 Platelet aggregation studies in these patients may only be moderately affected, confounding laboratory diagnosis. Platelet electron microscopy demonstrates absence of alpha granules. Patients with GPS can develop myelofibrosis and splenomegaly.5
Paris-Trousseau thrombocytopenia is a disorder that presents with mild bleeding tendency with variable thrombocytopenia, abnormal giant alpha-granules in platelets, and dysmegakaryopoiesis. The original description was in patients with deletions in 11q leading to Jacobsen syndrome. It was later recognized that the platelet phenotype is due to dysfunctional FLI1 and FLI1 homozygous mutations in absence of 11q, leading to the same phenotype.6
Hermansky-Pudlak syndrome (HPS) is an autosomal recessive disorder caused by mutations in one of ten different genes that lead to platelet dense granule deficiency. These mutations affect melanosome formation and cellular trafficking in other organs and also lead to oculocutaneous albinism, neutropenia, pulmonary fibrosis, and granulomatous colitis.7 Molecular confirmation of the particular subtype is important as the non-hematologic manifestations are part of some types and not others, and hence identification of the particular mutation is helpful in prognostication and establishing a plan for surveillance of end organ damage.8
INHERITED THROMBOCYTOPENIA
Inherited thrombocytopenia is characterized by a reduction in platelet number with or without other associated non-hematologic conditions. From a diagnostic perspective, classification according to platelet size—small, normal, or large—can help guide the diagnostic process. Platelet size can be estimated through mean platelet volume (MPV), which is an automated measure available in most commercial blood counters, or through direct observation and measurement on a peripheral smear.9 Misdiagnosis of inherited thrombocytopenia as autoimmune thrombocytopenia (ITP) can result in inappropriate therapies and inadequate surveillance for additional medical complications. There are more than twenty-five different molecular defects described leading to inherited thrombocytopenia. Some, such as MYH9-related disorders (MYH9-RD), can affect up to one in 300,000 individuals, while others have been described only in one or two families.
Thrombocytopenia with large platelet size
The MYH9-related disorders encompass a group of macrothrombocytopenia with or without deafness, renal failure, cataracts, and transaminitis that can present with the pathognomonic finding of Döhle-like bodies within the cytoplasm of neutrophils. They were previously described as the May-Hegglin anomaly, Fechtner syndrome, Sebastian syndrome, and Epstein syndrome, yet they all result from autosomal dominant mutations in the cytoskeletal protein MYH9.10 The type of mutation and area of the protein affected correlates with the phenotype, both at the ultrastructural level (type of inclusions seen) and the clinical phenotype (type of end organ damage associated); hence, molecular characterization can be an important tool in prognostication and follow-up of the affected families.11
Thrombocytopenia with normal platelet size
Non-syndromic thrombocytopenias with increased risk of hematologic malignancy are associated with mutations in RUNX1, ANKRD26 and ETV6, which lead to thrombocytopenia with mild to moderate bleeding and no other syndromic associations.12-14 They all have a high risk of evolution to myeloid neoplasms (acute myeloid leukemia, myelodysplastic syndrome, chronic myeloid leukemia, and chronic myelomonocytic leukemia), and in the case of ETV6, predisposition to acute lymphoblastic leukemia.12-14 RUNX1 germline mutations can present with decreased delta storage granules and more significant bleeding.15 Some abnormalities in platelet aggregation, platelet electron transmission electron microscopy, and surface glycoprotein expression have been described; however, they are not diagnostic and confirmation requires molecular techniques.16
Thrombocytopenia with small platelet size
Wiskott-Aldrich syndrome (WAS)-related disorders and X-linked thrombocytopenia affect males almost exclusively. They comprise a spectrum of disorders with a wide range of clinical manifestations. The specific phenotype may be related to the location and nature of the genetic variant as well as the amount of residual protein expression.17 Patients typically present with profound thrombocytopenia, eczema, and recurrent infections and are also predisposed to autoimmune disorders and lymphoma.18
DIAGNOSTICS AND THERAPEUTICS
Due to the heterogeneity of these disorders and the diversity of phenotypes, a careful history and physical examination should always be performed. A broad differential diagnosis should be considered, particularly in differentiating immune or acquired forms from inherited thrombocytopenia. Initial diagnostic evaluation includes a complete blood count with a peripheral smear, followed by assays to assess the coagulation cascade and platelet function. Advanced diagnostic techniques, including platelet aggregation studies or genetic evaluation, often require specialized referral centers.19
Treatment of inherited platelet disorders will vary depending on severity of disease. The scope and scale of treatment should be personalized for each patient. At a minimum, patient education and counseling is recommended following diagnosis. Patients should be made aware of primary self-care options if mild bleeding were to occur as well as exposure to drugs that may impact platelet function (that is, aspirin).
For more severe cases, desmopressin (DDAVP) or antifibrinolytics may be used to control mild bleeding episodes, while platelet transfusion remains a primary option to address severe bleeding or prepare patients for surgeries with a high bleeding risk, such a neurosurgery.20 In some instances, such as in patients with GT, alloimmunization to platelet antigens occurs, and hemostatic support has to be provided with activated clotting factor concentrates.
Bone marrow transplant has been successful in treating inherited platelet disorders in highly selected cases, particularly those that have associated bone marrow failure or immunodeficiency as an associated feature.21 However, due to the high associated transplant morbidity and mortality, transplant is not widely recommended.
Gene therapy and gene-editing technologies are promising options currently under investigation that could greatly benefit patients with IPDs in the future.18
REFERENCES
- Faioni EM, Razzari C, Zulueta A, et al. Bleeding diathesis and gastro-duodenal ulcers in inherited cytosolic phospholipase-A2 alpha deficiency. Thromb Haemost. 2014;112(6):1182-1189.
- Lopez JA, Andrews RK, Afshar-Kharghan V, et al. Bernard-Soulier
syndrome. Blood. 1998;91(12):4397-4418. - Solh T, Botsford A, Solh, M. Glanzmann’s thrombasthenia: pathogenesis, diagnosis, and current and emerging treatment options. J Blood Med: 2015;6:219-227.
- Nurden AT, Nurden P. Should any genetic defect affecting alpha-
granules in platelets be classified as gray platelet syndrome? Am J Hematol. 2016;91(7):714-718. - Nurden AT, Nurden P. Inherited thrombocytopenias. Haematologica. 2007;92(9):1158-1164.
- Favier R, Jondeau K, Boutard, P, et al. Paris-Trousseau syndrome:
clinical, hematological, molecular data of ten new cases. Thromb Haemost. 2003:90(5):893-897. - D’Andrea G, Chetta M, Margaglione M. Inherited platelet disorders: thrombocytopenias and thrombocytopathies. Blood Transfus. 2009;7(4):278-292.
- Botero JP, Chen D, Majerus JA, et al. Hermansky-Pudlak syndrome subtype 5 (HPS-5) novel mutation in a 65 year-old with oculocutaneous hypopigmentation and mild bleeding diathesis: the importance of recognizing a subtle phenotype. Platelets. 2017;1-4. doi:10.1080/09537104.2017.1361019.
- Noris P, Klersy C, Zecca M, et al. Platelet size distinguishes between
inherited macrothrombocytopenias and immune thrombocytopenia. J Thromb Haemost. 2009;7(12):2131-2136. - Savoia A, Pecci A. MYH9-Related Disorders. In MP Adam, HH Ardinger, RA Pagon, et al. (eds.), GeneReviews. Seattle WA.
- Pecci A, Klersy C, Gresele P, et al. MYH9-related disease: a novel prognostic model to predict the clinical evolution of the disease based on
genotype-phenotype correlations. Hum Mutat. 2014;35(2):236-247. - Melazzini F, Palombo F, Balduini A, et al. Clinical and pathogenic features of ETV6-related thrombocytopenia with predisposition to acute lymphoblastic leukemia. Haematologica. 2016;101(11):1333-1342.
- Noris P, Favier R, Alessi MC, et al. ANKRD26-related thrombocytopenia and myeloid malignancies. Blood. 2013;122(11):1987-1989.
- Song WJ, Sullivan MG, Legare RD, et al. Haploinsufficiency of CBFA2 causes familial thrombocytopenia with propensity to develop acute myelogenous leukaemia. Nat Genet. 199;23(2):166-175.
- Perez Botero J, Chen D, Cousin MA, et al. Clinical characteristics and platelet phenotype in a family with RUNX1 mutated thrombocytopenia. Leuk Lymphoma. 2017;58(8):1963-1967.
- Perez Botero J, Chen D, He R, et al. Clinical and laboratory characteristics in congenital ANKRD26 mutation-associated thrombocytopenia: a
detailed phenotypic study of a family. Platelets. 2016;27(7):712-715. - Drachman JG. Inherited thrombocytopenia: when a low platelet count does not mean ITP. Blood. 2004;103(2):390-398.
- Hacein-Bey AS, Gaspar HB, Blondeau J, et al. Outcomes following gene therapy in patients with severe Wiskott-Aldrich syndrome. JAMA. 2015; 313(15):1550-1563.
- Perez Botero J, Warad DM, He R, et al. Comprehensive platelet phenotypic laboratory testing and bleeding history scoring for diagnosis of suspected hereditary platelet disorders: a single-institution experience. Am J Clin Pathol. 2017;148(1):23-32.
- Orsini S, Noris P, Bury L, et al. Bleeding risk of surgery and its prevention in patients with inherited platelet disorders. Haematologica. 2017;102(7):1192-1203.
- Mullen CA, Anderson KD, Blaese RM. Splenectomy and/or bone marrow transplantation in the management of the Wiskott-Aldrich syndrome: long-term follow-up of 62 cases. Blood. 1993;82(10):2961-2966.
Juliana Perez Botero, MD, serves as an Associate Medical Director at BloodCenter of Wisconsin and Assistant Professor of Medicine at Medical College of Wisconsin. Dr. Perez Botero completed her training in Internal Medicine and Hematology and Oncology at the Mayo Clinic in Rochester, MN. She specializes in non-malignant hematologic disorders, including inherited and acquired bleeding and clotting disorders with a special focus on phenotype-genotype correlation in patients with familial platelet disorders.